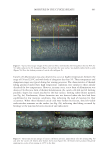
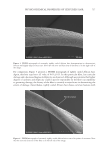
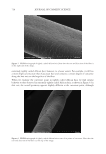
702 JOURNAL OF COSMETIC SCIENCE aqueous solution at neutral pH, tyrosine exhibits two absorption bands at wavelengths of about 220 and 275 nm, which are assigned to π–π* transitions (18). Upon absorption of a photon, the amino acid is excited to the first singlet excited state. The subsequent pathways are shown in Scheme 1. Once formed, tyrosyl radicals can undergo a wide variety of reactions. Two radicals can combine through either a C–C linkage or a C–O linkage to form dityrosine (19) (Scheme 2). Initial experiments were performed on tyrosine itself, but its photochemistry was not amenable to study. In solution, any tyrosyl radicals formed were too short-lived to be studied, but as a solid, the radicals that formed persisted for many hours. These data highlighted the importance of the molecular environment for photochemistry pathways. This phenomenon has also been seen in hair studies in which protein degradation was found to be highly dependent on relative humidity (23). The aim in this work was to create a system with a restricted tyrosine environment that still allowed for some molecular mobility. Toward this end, a series of amphiphilic PEG– Tyr block copolymers were used (Figure 1) (14). The hydrophilicity of the PEG block and the hydrophobicity of the Tyr block cause the copolymers to self-assemble into kinetically trapped, stable colloidal structures upon dissolution in water. Upon heating, the solutions spontaneously convert into more thermodynamically stable gels (24). Fluorescence spectroscopy was used to monitor the loss of tyrosine and formation of dityrosine. Samples were irradiated in 1 cm-path-length quartz cuvettes. Initial experiments used the PEG 5000 –Tyr 10 polymer irradiated for 3 and 24 h, with fluorescence of the sample measured relative to that of a control sample prepared at the same time but kept in the Scheme 2. Formation of dityrosine by the combination of two tyrosyl radicals. Figure 1. Structure of PEG–Tyr block copolymers.
703 UV OXIDATION dark. The degradation measured after the 3 h exposure period was 16%. Over a 24 h exposure period, 52% degradation was observed (Figure 2). Experiments were then performed with iron chloride salts to understand the role of redox metals in ROS formation. Increased tyrosine degradation was found, but iron levels greater than 100 ppm were needed because of the lower solubility of chloride salts in the PEG– Tyr micelles. Thus, two hydrophobic iron(III) salts that can localize into the polymer micelles were used, namely, iron(III) stearate [Fe(stearate) 3 ] and iron(III) acetylacetonate [Fe(acac) 3 ]. The percentage tyrosine degradation was measured over 24 h as a function of added Fe(stearate) 3 . Even at very low metal concentration (0.1 μg/g), the degradation was found to be significantly higher than in the metal-free systems (Figure 3). This iron salt 3 h 24 h 0 10 20 30 40 50 60 70 80 90 100 Figure 2. Tyrosine degradation upon UV exposure of a 10 mg/mL PEG5000–Tyr10 aqueous micellar solution, measured in terms of the decrease in tyrosine fluorescence relative to that of a control sample. Figure 3. Effect of the Fe(stearate)3 concentration (expressed as the Fe:Tyr ratio) on tyrosine degradation in PEG2000–Tyr5 micellar systems at 2 mg/mL, measured in terms of the decrease in tyrosine fluorescence over 24 h of UV exposure. yrfuoescenceost T %
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)