202 JOURNAL OF COSMETIC SCIENCE
In this paper, to obtain safer and more environmentally friendly polyphenol-based hair
dyes, the effects of different parameters (i.e., nine polyphenols, Fe(II) or Fe(III), pH, time)
on the coloration of the dyed gray hair were investigated. When ethanolamine (EA) was
used as an alkalizing substance to open the hair cuticle, natural white hair could be dyed
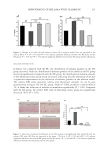
by using the complex of GA and Fe(II), and the dyed hair fibers showed good durability in
washing with the aid of dopamine hydrochloride (DA) (Figure 1).
MATERIALS AND METHODS
MATERIALS
GA, DA, Tannic acid (TA), Caffeic acid (CA), Resorcinol (RE), ascorbic acid, sodium
benzoate, glycerol, and tween-80 were purchased from Shanghai Aladdin Biochemical
Technology (Shanghai, China). 3, 4-Dihydroxybenzaldehyde (Protocatechualdehyde PA),
Catechin hydrate (CH), Tris (hydroxymethyl)aminomethane (Tris), ethanolamine (EA),
oleic acid, and sodium dodecyl sulfate were purchased from Shanghai Macklin Biochemical.
(Shanghai, China). Salicylic acid (SA) from Yuanye Bio-Technology (Shanghai, China).
Pyrogallol (PG) and Ethanol absolute from Energy Chemical (Shanghai, China). Ferrous
chloride tetrahydrate from Meyer (Shanghai, China). 1,10-Phenanthroline from Leyan
Technologies (Shanghai, China). Sodium acetate from Jinshan Chemical Test (Chengdu,
China). Hydroxylamine hydrochloride, Anhydrous sodium sulfite, and isopropanol from
Tianjin Kermel Chemical Reagent Co. (Tianjin, China). Propylene glycol from Tianjin
Fuyu Fine Chemical Co. (Tianjin, China). Sodium thiosulfate from Youpu (Tianjin, China).
Commercial white hair (9° platinum blonde) was purchased from YNE (China), human
natural white hair (non-commercial) samples (provided by volunteers), and pure water
(ELGA) prepared by PURELAB Flex ultrapure water system was used for the experiments.
Figure 1. Schematic diagram of polyphenol-metal coordination structure compound dopamine dyeing
natural white hair.
In this paper, to obtain safer and more environmentally friendly polyphenol-based hair
dyes, the effects of different parameters (i.e., nine polyphenols, Fe(II) or Fe(III), pH, time)
on the coloration of the dyed gray hair were investigated. When ethanolamine (EA) was
used as an alkalizing substance to open the hair cuticle, natural white hair could be dyed
by using the complex of GA and Fe(II), and the dyed hair fibers showed good durability in
washing with the aid of dopamine hydrochloride (DA) (Figure 1).
MATERIALS AND METHODS
MATERIALS
GA, DA, Tannic acid (TA), Caffeic acid (CA), Resorcinol (RE), ascorbic acid, sodium
benzoate, glycerol, and tween-80 were purchased from Shanghai Aladdin Biochemical
Technology (Shanghai, China). 3, 4-Dihydroxybenzaldehyde (Protocatechualdehyde PA),
Catechin hydrate (CH), Tris (hydroxymethyl)aminomethane (Tris), ethanolamine (EA),
oleic acid, and sodium dodecyl sulfate were purchased from Shanghai Macklin Biochemical.
(Shanghai, China). Salicylic acid (SA) from Yuanye Bio-Technology (Shanghai, China).
Pyrogallol (PG) and Ethanol absolute from Energy Chemical (Shanghai, China). Ferrous
chloride tetrahydrate from Meyer (Shanghai, China). 1,10-Phenanthroline from Leyan
Technologies (Shanghai, China). Sodium acetate from Jinshan Chemical Test (Chengdu,
China). Hydroxylamine hydrochloride, Anhydrous sodium sulfite, and isopropanol from
Tianjin Kermel Chemical Reagent Co. (Tianjin, China). Propylene glycol from Tianjin
Fuyu Fine Chemical Co. (Tianjin, China). Sodium thiosulfate from Youpu (Tianjin, China).
Commercial white hair (9° platinum blonde) was purchased from YNE (China), human
natural white hair (non-commercial) samples (provided by volunteers), and pure water
(ELGA) prepared by PURELAB Flex ultrapure water system was used for the experiments.
Figure 1. Schematic diagram of polyphenol-metal coordination structure compound dopamine dyeing
natural white hair.