212 JOURNAL OF COSMETIC SCIENCE
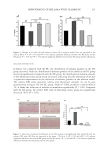
the dye molecules, making them less susceptible to washing and enhancing the washing
resistance of dyeing (Figure 7C). We determined that permanent hair dyeing could be
achieved using GA-Fe(II)+DA hair dye. In addition, the introduction of DA resulted in
a subsequent increase in the color difference value of the hair, i.e., a better dyeing effect.
In addition, we did a tensile test on dyed hair versus hair without dyeing treatment, and
according to the results in Figure 7D, the dyed hair was able to withstand greater force
within a period of strain value (approximately 32%), and the hair broke when this strain
value was exceeded. However, both dyed and undyed hair can withstand similar stress
values, which means that the dyed hair can withstand the same force as undyed natural
white hair. However, it can withstand less strain in the stress range, i.e., the dyed hair
has slightly poorer tensile resistance, which we speculate is caused by the unfolded hair
scales not closing in time by ethanolamine. Nevertheless, over time, the unfolded hair
scales gradually close, and through oxidation with the deposited dopamine in the cortical
layer, they self-polymerize into polymeric polydopamine. This polymeric substance forms
an adhesive film within the cortical layer, encapsulating and securing the materials within
the hair to prevent loss. Moreover, it enhances the tensile properties of the hair.
CONCLUSIONS
In this study, we developed a method for dyeing natural white hair by creating a complex
formed through the interaction of polyphenols with metals, followed by the addition of
dopamine to formulate a complex hair dye. With the help of the alkaline environment of
Figure 7. (A) Van der Waals ball model of DA, GA, and GA-Fe(II). (B) Ball-and-stick model of DA, GA with
GA-Fe(II). (C) ΔE values of GA-Fe(II) and GA-Fe(II)+DA for washing resistance test. (D) Stress-strain curves
of natural white hair (untreated) and GA-Fe(II)+DA dyed hair.
the dye molecules, making them less susceptible to washing and enhancing the washing
resistance of dyeing (Figure 7C). We determined that permanent hair dyeing could be
achieved using GA-Fe(II)+DA hair dye. In addition, the introduction of DA resulted in
a subsequent increase in the color difference value of the hair, i.e., a better dyeing effect.
In addition, we did a tensile test on dyed hair versus hair without dyeing treatment, and
according to the results in Figure 7D, the dyed hair was able to withstand greater force
within a period of strain value (approximately 32%), and the hair broke when this strain
value was exceeded. However, both dyed and undyed hair can withstand similar stress
values, which means that the dyed hair can withstand the same force as undyed natural
white hair. However, it can withstand less strain in the stress range, i.e., the dyed hair
has slightly poorer tensile resistance, which we speculate is caused by the unfolded hair
scales not closing in time by ethanolamine. Nevertheless, over time, the unfolded hair
scales gradually close, and through oxidation with the deposited dopamine in the cortical
layer, they self-polymerize into polymeric polydopamine. This polymeric substance forms
an adhesive film within the cortical layer, encapsulating and securing the materials within
the hair to prevent loss. Moreover, it enhances the tensile properties of the hair.
CONCLUSIONS
In this study, we developed a method for dyeing natural white hair by creating a complex
formed through the interaction of polyphenols with metals, followed by the addition of
dopamine to formulate a complex hair dye. With the help of the alkaline environment of
Figure 7. (A) Van der Waals ball model of DA, GA, and GA-Fe(II). (B) Ball-and-stick model of DA, GA with
GA-Fe(II). (C) ΔE values of GA-Fe(II) and GA-Fe(II)+DA for washing resistance test. (D) Stress-strain curves
of natural white hair (untreated) and GA-Fe(II)+DA dyed hair.