239 IMPROVEMENT OF MELASMA WITH GLABRIDIN
and the melanin content in nonmelasma areas. However, no clear differences in the melanin
content were found between the two groups. The uneven color of melasma may contribute
to this finding, as subtle differences in the test values may not be accurately reflected in the
treatment outcome. At the same time, it was relatively difficult to remove melanin, due
to its deep location in the epidermis. It seemed that only addition of glabridin displayed
limited effects. The combination with other whitening ingredients was recommend to
achieve better whitening effects.
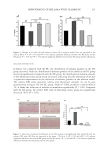
The skin color modulated by the transfer of melanin synthesized by melanocytes was
embedded by the melanosome to neighboring keratinocytes. The 3D reconstructed
pigmented skin model composed of human melanocytes in the basal layer of multilayered
epidermal keratinocytes was used to detect the changes of melanin content. Hence, the
distribution of melanin in the different skin layers can be well recognized by its brown-
colored pigmentation for the 3D pigmented skin model. The inhibitory effects on
melanogenesis of the whitening serum containing glabridin was also evaluated by 3D skin
model. The whitening serum effectively improved the apparent chromaticity of the melanin
model, increased the L* value, and regulated the content and distribution of melanin.
CONCLUSION
The whitening serum containing glabridin effectively improved the apparent chromaticity
of the melanin model, increased the L* value, and regulated the content and distribution
of melanin. A 56-day clinical experiment showed that glabridin effectively improved
the skin glossiness and ITA value in both melasma and nonmelasma areas. Meanwhile,
a remarkably reduced melasma area proportion and melanin content was observed in
melasma areas and nonmelasma areas, respectively. In summary, these results suggest that
a formula containing glabridin could effectively improve pigmentation in 3D skin models
and volunteers with epidermal melasma.
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding
author upon reasonable request.
ACKNOWLEDGMENTS
The study was supported by the Zhejiang Osmum Biological Co., Ltd, Huzhou, China.
CONFLICT OF INTEREST
The authors declare that they have no competing interests.
AUTHOR CONTRIBUTIONS
Gang Huo, Liping Du, and Ping Ma designed the experiments and wrote the paper. Ying
Zhou performed the experiments. Xinfen Cai analyzed the data.
and the melanin content in nonmelasma areas. However, no clear differences in the melanin
content were found between the two groups. The uneven color of melasma may contribute
to this finding, as subtle differences in the test values may not be accurately reflected in the
treatment outcome. At the same time, it was relatively difficult to remove melanin, due
to its deep location in the epidermis. It seemed that only addition of glabridin displayed
limited effects. The combination with other whitening ingredients was recommend to
achieve better whitening effects.
The skin color modulated by the transfer of melanin synthesized by melanocytes was
embedded by the melanosome to neighboring keratinocytes. The 3D reconstructed
pigmented skin model composed of human melanocytes in the basal layer of multilayered
epidermal keratinocytes was used to detect the changes of melanin content. Hence, the
distribution of melanin in the different skin layers can be well recognized by its brown-
colored pigmentation for the 3D pigmented skin model. The inhibitory effects on
melanogenesis of the whitening serum containing glabridin was also evaluated by 3D skin
model. The whitening serum effectively improved the apparent chromaticity of the melanin
model, increased the L* value, and regulated the content and distribution of melanin.
CONCLUSION
The whitening serum containing glabridin effectively improved the apparent chromaticity
of the melanin model, increased the L* value, and regulated the content and distribution
of melanin. A 56-day clinical experiment showed that glabridin effectively improved
the skin glossiness and ITA value in both melasma and nonmelasma areas. Meanwhile,
a remarkably reduced melasma area proportion and melanin content was observed in
melasma areas and nonmelasma areas, respectively. In summary, these results suggest that
a formula containing glabridin could effectively improve pigmentation in 3D skin models
and volunteers with epidermal melasma.
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding
author upon reasonable request.
ACKNOWLEDGMENTS
The study was supported by the Zhejiang Osmum Biological Co., Ltd, Huzhou, China.
CONFLICT OF INTEREST
The authors declare that they have no competing interests.
AUTHOR CONTRIBUTIONS
Gang Huo, Liping Du, and Ping Ma designed the experiments and wrote the paper. Ying
Zhou performed the experiments. Xinfen Cai analyzed the data.