210 JOURNAL OF COSMETIC SCIENCE
solid determination. Figure 6D shows the FR-IT spectra of dopamine hydrochloride, GA,
ferrous chloride tetrahydrate, GA-Fe(II) solution, and GA-Fe(II)+DA solution. In the
FR-IT spectra of GA-Fe(II)+DA, we found that 1,611 cm−1 and 1,522 cm−1 are benzene
ring skeleton vibrational absorption peaks. Additionally, a peak at 1,286 cm−1 indicates
carbon-oxygen single bond stretching vibrations. Importantly, the strong absorption
peak between 1,100 cm−1 and 1,000 cm−1, consistent with ferrous chloride tetrahydrate,
confirms the complete formation of the GA-Fe(II) complex structure. In addition, the
spectrum of GA-Fe(II)+DA showed no bending vibration peaks of amine and stretching
vibration peaks of carbon and nitrogen bonds, which proved that DA was not involved in
the complexation in the coordination structure of GA-Fe(II), as DA was removed during
the freeze-drying process.
Although GA-Fe(II)+DA hair dye can dye commercial white hair, the dye molecules cannot
penetrate the cortical layer of hair through the protective layer of hair scales of natural white
hair without the help of external forces. This is because the maximum critical molecular
size that can be allowed to pass through the protective layer of hair scales is 1.4 nm. Figure
7A shows the van der Waals sphere model for calculating the molecular sizes of GA, DA,
and GA-Fe(II). The results show that the molecular size of GA is 0.965 nm ≤ 1.4 nm, and
when GA forms a complex with Fe(II), the molecular size increases to ≥ 1.4 nm. The larger
the molecular volume of the complex, the larger the molecular size, making it impossible
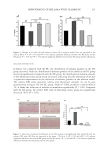
Figure 5. (A) Time screening of GA-Fe(II)+DA impregnated natural gray hair. (B) Molar ratio screening of
GA to Fe(II). (C), (D) pH screening of GA-Fe(II)+DA.
solid determination. Figure 6D shows the FR-IT spectra of dopamine hydrochloride, GA,
ferrous chloride tetrahydrate, GA-Fe(II) solution, and GA-Fe(II)+DA solution. In the
FR-IT spectra of GA-Fe(II)+DA, we found that 1,611 cm−1 and 1,522 cm−1 are benzene
ring skeleton vibrational absorption peaks. Additionally, a peak at 1,286 cm−1 indicates
carbon-oxygen single bond stretching vibrations. Importantly, the strong absorption
peak between 1,100 cm−1 and 1,000 cm−1, consistent with ferrous chloride tetrahydrate,
confirms the complete formation of the GA-Fe(II) complex structure. In addition, the
spectrum of GA-Fe(II)+DA showed no bending vibration peaks of amine and stretching
vibration peaks of carbon and nitrogen bonds, which proved that DA was not involved in
the complexation in the coordination structure of GA-Fe(II), as DA was removed during
the freeze-drying process.
Although GA-Fe(II)+DA hair dye can dye commercial white hair, the dye molecules cannot
penetrate the cortical layer of hair through the protective layer of hair scales of natural white
hair without the help of external forces. This is because the maximum critical molecular
size that can be allowed to pass through the protective layer of hair scales is 1.4 nm. Figure
7A shows the van der Waals sphere model for calculating the molecular sizes of GA, DA,
and GA-Fe(II). The results show that the molecular size of GA is 0.965 nm ≤ 1.4 nm, and
when GA forms a complex with Fe(II), the molecular size increases to ≥ 1.4 nm. The larger
the molecular volume of the complex, the larger the molecular size, making it impossible
Figure 5. (A) Time screening of GA-Fe(II)+DA impregnated natural gray hair. (B) Molar ratio screening of
GA to Fe(II). (C), (D) pH screening of GA-Fe(II)+DA.