208 JOURNAL OF COSMETIC SCIENCE
Consequently, dye molecules could easily penetrate the hair cortical layer and achieve
successful dyeing.
In contrast, natural white hair retained intact hair scales and an undisturbed hair structure,
preventing dye molecules from directly passing through the scales into the hair cortex
for effective dyeing. Therefore, we prepared an ethanolamine emulsion (Figure 3E) in
an alkaline environment to unfold the hair scales of natural white hair so that the dye
molecules of GA-Fe(II)+DA hair dye could directly pass through the hair scales into
the hair cortex for deposition to achieve dyeing of natural white hair (Figure 3D). SEM
images of natural white hair treated with ethanolamine emulsion and natural white hair
dyed with GA-Fe(II)+DA hair dye after unfolding the hair scales with ethanol emulsion
showed (Figure 3I, J) that the hair surface was smooth without the presence of dye molecule
adhesion films. This indicated that both dye molecules and dopamine entered the hair
cortical layer. Furthermore, the hair scales in the epidermal layer of dyed natural white
hair exhibited no debonding, layering, or flexing, and the hair surface remained smooth
and structurally intact. These findings were consistent with the results obtained from
SEM images of untreated natural white hair. They indicated that both the ethanolamine
emulsion and the GA-Fe(II)+DA hair dye did not adversely affect the hair structure and
that the intact hair scales helped retain moisture in the hair more effectively.30
The raw materials used in both the ethanolamine emulsion and hair dye are cosmetic
ingredients approved for use by the China Food &Drug Administration. In addition, ferrous
ions are relatively non-toxic compared to other heavy metals. According to the regulatory
guidelines of the Korean Ministry of Food and Drug Safety, a certain concentration of
ferrous ions is allowed to be used on human hair and scalp.13 Therefore, GA-Fe(II)+DA
hair dye may be a potential safer alternative to traditional hair dyes.
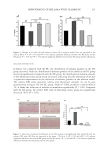
In addition, the effects of ethanolamine solution pH (Figures 4A, 4B, 4C), ethanolamine
concentration (Figure 4D), and the time of ethanolamine soaking natural gray hair
(Figure 4E) on hair scaling, and consequently, the hair coloring effect, were evaluated.
Optimal conditions for achieving the desired dyeing effect with ethanolamine were
determined based on the ΔE value. The best results were observed when the pH of the
ethanolamine solution was within the range of 9.2–9.4, with the most effective dyeing of
natural white hair occurring in this pH range. Deviating from this pH range resulted in a
decrease in the color difference during dyeing. It’s important to note that excessively high
or low pH levels can be detrimental to human hair and the scalp. Thus, maintaining a pH
of 9.2–9.4 is considered more suitable for this process.
Moreover, soaking natural gray hair in ethanolamine for 1 hour proved to be the most
reasonable duration in the context of the hair coloring industry. Lastly, an ethanolamine
concentration of 20%,was found to be the most effective for coloring natural gray hair.
We also screened the effects of GA-Fe(II)+DA impregnation time on natural gray hair
(Figure 5A), the molar ratio of GA to Fe(II) (Figure 5B), and the pH of GA-Fe(II)+DA
(Figure 5C-D) on the effect of natural gray hair coloring. Our findings revealed that when
the molar ratio of GA to Fe(II) was 1:5, the time of GA-Fe(II)+DA impregnating natural
white hair was 2 hours, and the pH of GA-Fe(II)+DA hair dye was 5.0–5.2. When the
pH of GA-Fe(II)+DA was screened, we found that if the pH of GA-Fe(II)+DA is alkaline,
Fe(II) in the solution will be precipitated as Fe(OH)
2 ,which cannot be prepared as a hair
dye. In addition, hair dyes with an acidic pH can effectively promote the closure of hair
scales and prevent the loss of hair coloring components in the hair. Of course, the dyeing
Consequently, dye molecules could easily penetrate the hair cortical layer and achieve
successful dyeing.
In contrast, natural white hair retained intact hair scales and an undisturbed hair structure,
preventing dye molecules from directly passing through the scales into the hair cortex
for effective dyeing. Therefore, we prepared an ethanolamine emulsion (Figure 3E) in
an alkaline environment to unfold the hair scales of natural white hair so that the dye
molecules of GA-Fe(II)+DA hair dye could directly pass through the hair scales into
the hair cortex for deposition to achieve dyeing of natural white hair (Figure 3D). SEM
images of natural white hair treated with ethanolamine emulsion and natural white hair
dyed with GA-Fe(II)+DA hair dye after unfolding the hair scales with ethanol emulsion
showed (Figure 3I, J) that the hair surface was smooth without the presence of dye molecule
adhesion films. This indicated that both dye molecules and dopamine entered the hair
cortical layer. Furthermore, the hair scales in the epidermal layer of dyed natural white
hair exhibited no debonding, layering, or flexing, and the hair surface remained smooth
and structurally intact. These findings were consistent with the results obtained from
SEM images of untreated natural white hair. They indicated that both the ethanolamine
emulsion and the GA-Fe(II)+DA hair dye did not adversely affect the hair structure and
that the intact hair scales helped retain moisture in the hair more effectively.30
The raw materials used in both the ethanolamine emulsion and hair dye are cosmetic
ingredients approved for use by the China Food &Drug Administration. In addition, ferrous
ions are relatively non-toxic compared to other heavy metals. According to the regulatory
guidelines of the Korean Ministry of Food and Drug Safety, a certain concentration of
ferrous ions is allowed to be used on human hair and scalp.13 Therefore, GA-Fe(II)+DA
hair dye may be a potential safer alternative to traditional hair dyes.
In addition, the effects of ethanolamine solution pH (Figures 4A, 4B, 4C), ethanolamine
concentration (Figure 4D), and the time of ethanolamine soaking natural gray hair
(Figure 4E) on hair scaling, and consequently, the hair coloring effect, were evaluated.
Optimal conditions for achieving the desired dyeing effect with ethanolamine were
determined based on the ΔE value. The best results were observed when the pH of the
ethanolamine solution was within the range of 9.2–9.4, with the most effective dyeing of
natural white hair occurring in this pH range. Deviating from this pH range resulted in a
decrease in the color difference during dyeing. It’s important to note that excessively high
or low pH levels can be detrimental to human hair and the scalp. Thus, maintaining a pH
of 9.2–9.4 is considered more suitable for this process.
Moreover, soaking natural gray hair in ethanolamine for 1 hour proved to be the most
reasonable duration in the context of the hair coloring industry. Lastly, an ethanolamine
concentration of 20%,was found to be the most effective for coloring natural gray hair.
We also screened the effects of GA-Fe(II)+DA impregnation time on natural gray hair
(Figure 5A), the molar ratio of GA to Fe(II) (Figure 5B), and the pH of GA-Fe(II)+DA
(Figure 5C-D) on the effect of natural gray hair coloring. Our findings revealed that when
the molar ratio of GA to Fe(II) was 1:5, the time of GA-Fe(II)+DA impregnating natural
white hair was 2 hours, and the pH of GA-Fe(II)+DA hair dye was 5.0–5.2. When the
pH of GA-Fe(II)+DA was screened, we found that if the pH of GA-Fe(II)+DA is alkaline,
Fe(II) in the solution will be precipitated as Fe(OH)
2 ,which cannot be prepared as a hair
dye. In addition, hair dyes with an acidic pH can effectively promote the closure of hair
scales and prevent the loss of hair coloring components in the hair. Of course, the dyeing