206 JOURNAL OF COSMETIC SCIENCE
A =+6.7356c3 0.0077 (eq. 1)
c
2 10c3 =(eq. 2)
m c2V
2 =(eq. 3)
m MFe M m0
1 =÷ × (eq. 4)
w m1 m m1 =-×100% ()÷
2 (eq. 5)
Among them, c
3
is used to measure the concentration of Fe(II) in the solution during
absorbance c
2
is the concentration of Fe(II) in the centrifuged supernatant after the reaction
m
2
is the mass of Fe(II) in the centrifuged supernatant after the reaction V is the reaction
volume, which is 10 mL m
1
is the total mass of Fe(II) in the reaction system m
0
is the
mass of weighed ferrous chloride tetrahydrate M
Fe
is the molar mass of iron (M
Fe
=56 g/
mol) M is the molar mass of ferrous chloride tetrahydrate (M =198.71 g/mol) w is the
chelation rate.
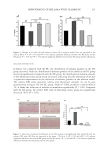
Among the color difference results of 10 polyphenols reacted with Fe (II) to prepare hair
dyeing solution to dye natural white hair, the color difference values of GA, PA, CH, and
CA were all above 60, indicating that these four materials had a better dyeing effect on
Figure 2. (A) Color difference value of polyphenol and Fe(II) stained natural white hair and (B) standard
curve.
Table I
Absorbance Value and Chelation Rate of Various Polyphenols and Fe(II)
Polyphenol m0 (g) A (mL/ mol/cm) c3 (mg/mL) m2 (mg) m1 (mg) w (%)
GA 0.2050 0.3517 0.0511 5.1100 57.7726 91.15
TA 0.2302 0.6199 0.0909 9.0900 64.8744 85.99
PA 0.2520 0.4892 0.0715 7.1500 71.0181 89.93
PG 0.2742 0.6644 0.0975 9.7500 77.2744 87.38
CH 0.1205 0.7068 0.1038 10.3800 33.9590 69.43
SA 0.1941 0.6633 0.0973 9.7300 71.0744 86.31
CA 0.2522 0.6535 0.0959 9.5900 54.7008 82.47
DA 0.2266 0.6031 0.0884 8.8400 63.8599 86.16
PDA 0.2286 0.6015 0.0882 8.8200 64.4423 86.31
A =+6.7356c3 0.0077 (eq. 1)
c
2 10c3 =(eq. 2)
m c2V
2 =(eq. 3)
m MFe M m0
1 =÷ × (eq. 4)
w m1 m m1 =-×100% ()÷
2 (eq. 5)
Among them, c
3
is used to measure the concentration of Fe(II) in the solution during
absorbance c
2
is the concentration of Fe(II) in the centrifuged supernatant after the reaction
m
2
is the mass of Fe(II) in the centrifuged supernatant after the reaction V is the reaction
volume, which is 10 mL m
1
is the total mass of Fe(II) in the reaction system m
0
is the
mass of weighed ferrous chloride tetrahydrate M
Fe
is the molar mass of iron (M
Fe
=56 g/
mol) M is the molar mass of ferrous chloride tetrahydrate (M =198.71 g/mol) w is the
chelation rate.
Among the color difference results of 10 polyphenols reacted with Fe (II) to prepare hair
dyeing solution to dye natural white hair, the color difference values of GA, PA, CH, and
CA were all above 60, indicating that these four materials had a better dyeing effect on
Figure 2. (A) Color difference value of polyphenol and Fe(II) stained natural white hair and (B) standard
curve.
Table I
Absorbance Value and Chelation Rate of Various Polyphenols and Fe(II)
Polyphenol m0 (g) A (mL/ mol/cm) c3 (mg/mL) m2 (mg) m1 (mg) w (%)
GA 0.2050 0.3517 0.0511 5.1100 57.7726 91.15
TA 0.2302 0.6199 0.0909 9.0900 64.8744 85.99
PA 0.2520 0.4892 0.0715 7.1500 71.0181 89.93
PG 0.2742 0.6644 0.0975 9.7500 77.2744 87.38
CH 0.1205 0.7068 0.1038 10.3800 33.9590 69.43
SA 0.1941 0.6633 0.0973 9.7300 71.0744 86.31
CA 0.2522 0.6535 0.0959 9.5900 54.7008 82.47
DA 0.2266 0.6031 0.0884 8.8400 63.8599 86.16
PDA 0.2286 0.6015 0.0882 8.8200 64.4423 86.31