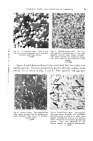
THE SOCIETY JOURNAL OF THE OF COSMETIC CHEMISTS This edition is published by THE SOCIETY OF COSMETIC CHEMISTS OF GREAT BRITAIN Publications Office: 54, Woodlands, London, N.W. 11. VOL. X JANUARY 1959 1 PIGMENTS, LAKES AND DYESTUFFS IN COSMETICS D. F. ANSTEAD, B.Sc., A.R.I.C.* A lecture delivered before the Society on 3rd Ja•mary 1958. General manufacturing details of dyestuffs and pigments together with their properties are dealt with. There is a summary of the requirements of colours in cosmetics as well as of the relevant legislation in Great Britain and North America. THE TITLE of this paper, as set out in the programme, •vould have been more descriptive of the actual contents if it had been prefixed with the word "organic" as it is intended to discuss dyestuffs and pigments of this type, only referring to the large group of inorganic pigments, when necessary to illustrate a point. It is appreciated that inorganic pigments are used to a large extent in the cosmetic industry but they would deserve a paper to themselves, so that the emphasis will be on the organic pigments and dyestuffs. It would be well to define as simply as possible the headings under which, for the purpose of this paper, they will be grouped. A dyestuff is defined as a material that imparts its colour to the full only in solution, the solvent being water or some other medium such as alcohol, oil, petroleum products, etc. There may be some action on the addition of the dyestuff solution to the material to be coloured which renders the dye- stuff either insoluble or partly soluble but in the first process it makes a true solution in the solvent. Three simple illustrations of this action are as follows: 1. A solution of the dyestuff in vater. The water evaporates leaving the dyestuff, from the practical angle, colourless. * D. F. Anstead, Ltd., Romford, Essex.
JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 2. A solution of the natural colouring matter Carmine in ammoniacai water. The ammonia evaporates leaving the insoluble lake behind. :3. When an aqueous solution of a basic dyestuff is added to certain silicates, an insoluble lake is precipitated (double decomposition). These points are made as it is important to appreciate that the addition of the colouring matter is not the end. The conditions with which it has to contend have a considerable bearing on its useful life. Organic Pigments It is not easy to divide this complex family into sections. Scientifically, they ought to be considered under their various chemical groups but, for our purpose, fairly simple headings can be made that will include most pigments, the headings being decided by the method of manufacture rather than by constitution. Lake Pigments A lake, in the broadest sense of the term, can be described as a soluble dyestuff or colouring matter rendered insoluble by some suitable means and precipitated on a base or sub-stratum. Such a definition implies three essential constituents: 1. Colouring matter, 2. precipitating agent, and 3. base or sub-stratum. In the third constituent the word "base" should not be confused with the chemical "base" but it is an accepted term in the colour-making industry and implies that it is there as an essential constituent of the lake and is not added for the purpose of diluting, extending or cheapening the lake pigment. In many cases this base fulfils a necessary and important function in the lake and its omission would give a product which would be useless as a pigmentary colour. The preparation of the base calls for a knowledge of the dyestuff to be laked and, in some cases, how it is to be used. To illustrate how the manufacturing process can affect the final product aluminium hydroxide, one of the most common bases, can be discussed. The material is usually shown as A12(OH)6 but we know this to be only part of the story as it also contains varying quantities of basic aluminium sulphate and in many cases the dyestuff would not precipitate unless this is present. The reaction can be shown thus: A12(SO4)3: 18H•O + 3NarCO3 -•AI•(OH)6 + 3Na•SO• + 3CO.• + 15H•O The reaction, as the sodium carbonate is run into the aluminium sulphate, is actually as follows: 1. Al•(SO4)• + Na•CO• + H•O -• A12(SO,)•(OH)• + Na•SO• + COs 2. AI•(SO•)•(OH)• + Na•CO• + H•O--Ai•(SO•)(OH)4 + Na•SO• + CO• 3. AI•(SO,)(OH), + Na•COa + H•O-- AI.•(OH)o + Na•SO• + CO2
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)