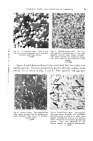
PIGMENTS, LAKES AND DYESTUFFS IN COSMETICS 3 Trying the experiment, it will be found that precipitation does not occur until over two-thirds of the sodium carbonate have been added. The following must also be considered: (a) That the A12(SO•)• can be substituted with any of the alums, (b) that potassium or ammonium carbonate can be used, and (c) that sodium, potassium or ammonium hydroxide can be used in place of the carbonates. All these factors modify the precipitate to a greater or lesser extent. As with all precipitations, contributing factors include: (i) the concentration of the solution, (ii) the rate of addition, (iii) aluminium salt into alkali or vice versa, (iv) the proportion of aluminium sulphate to soda ash which can be varied within certain limits, and (v) the temperature of precipitation, a factor of particular importance. The amount of basic aluminium sulphate left in the finished base depends to a large extent on the temperature. Cold, dilute solutions favour the retention of the basic aluminium sulphate and give very hard, transparent lakes, slow in settling and difficult to wash free from salts. Hot, concentrated solutions give flocculent bases, easily washed free from salts and containing less basic aluminium sulphate. The above factors all affect the properties of the alumina hydrate and consequently, the ease of precipitation of the dyestuff and the shade of the finished lake. In the precipitation of acid dyestuffs barium chloride is used most fre- quently, being added after addition of the dyestuff. From the above you will appreciate that the barium chloride will react with any retained basic aluminium sulphate thereby precipitating barium sulphate, the amount dependent on the proportion of basic aluminium sulphate left in the base. This is, in fact, the method of preparation of another well-known base "alumina-blanc fixe" where the alumina hydrate is not washed free from salts and consequently the dyestuff is precipitated on to a mixed base of alumina-barium sulphate. This base differs from alumina by giving opalescent lakes. The importance of the presence of a base can be well illustrated with the laking of the dyestuff "Ponceau R". Struck out with barium chloride without a base, the lake would be hard, dull and low in staining power. If alumina hydrate is present with the dyestuff before precipitation, a brilliant lake, soft in texture and high in staining power will result. Madder lake can also be given as an example and here it is found that not only is a suitably prepared base essential to a satisfactory colour but calcium, the phosphate group and turkey red oil must be introduced to obtain the softest and strongest lake.
4 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS The natural colouring matters are mostly used in lake form. The familiar Carmine which is obtained from the insect Coccus Cactii (Cochineal) is mainly the aluminium salt of carminic acid formed in the presence of alumina hydrate. Other natural colouring matters such as Brazil Wood and Lima Wood, which make Rose Pink, and Quercitron Bark and Persian Berries, which make Dutch Pink, are similar compounds. These are mentioned as interest is being shown in them arising from recent regulations governing the use of the natural colours in foodstuffs. Coal-tar dyestuffs used in lakes generally fall into three types: 1. Acid, 2. basic, and 3. adjective. In the first group, the colour complex is contained in the acid part of the compound and they are usually found in the form of their sodium or potassium salts. Their laking can be shown thus: Sodium dyate + Al•(SO4)• -+ A1 dyate + Na•SO4 soluble acid dyestuff + precipitant -+ insoluble lake. With the second group, the basic dyestuffs, the colour complex is contained in the basic part of the compound and they usually appear as the chloride, or sulphate. This may be shown thus: Dye sulphate + Sodium benzoate -- dye benzoate + Na•SO• Soluble basic dye + precipitant -+ insoluble lake. With the third group, the adjective dyestuffs, those yielding lakes are mainly of the alizarin type a typical example being 1:2: dihydroxyanthraquinone. Here the colourless alizarin is developed in the presence of a mordant. The most important member (above) yields crimson lakes but scarlet and maroon lakes can be prepared from various types of increasingly hydroxy- lated alizarin and by variations in the mordant. A minor class not detailed above is prepared from the vat dyestuff by reduction of the dyestuff to the leuco compound and subsequent oxidation in the presence of a base. The lakes thus formed, although they are renowned for their extreme fastness to light when used for textiles, are not particularly satisfactory as they do not show this property to the same extent when converted into pigment lakes. Pigment Dyestuffs are probably the most important division and one which contains the greatest number of different pigments embracing most of the important red and yellow colours. They are mainly of the azo type, containing the characteristic -- N = N -grouping which is formed in the process of manufacture from the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)