PIGMENTS, LAKES AND DYESTUFFS IN COSMETICS coupling of a primary and secondary component. When the result is a soluble dyestuff, as would be obtained from the coupling aniline + 2. naphthol ½:8. disulphonic acid --• Orange G the dyestuff is salted out and, for the purpose of this paper, would be con- sidered as an "acid dyestuff." When the end products are more or less insoluble they are described as pigment dyestuffs. The pigment dyestuffs fall into two distinctive groups: 1. Those that are pigments in themselves and require no further pro- cessing. 2. Those that initially form the sodium or potassium salt and can be laked, i.e. they behave as acid dyestuffs but require no base or sub- stratum for the full development of their pigmentary properties. The compounds are usually laked with a heavy metal salt such as barium, calcium or strontium, and are described as "pigment dyestuff toners." These toners can be used in their primary form as sodium salts, being insoluble in oil and very sparingly soluble in water. Laking with the heavy metal usually gives increased fastness to light as well as modi- fying the shade. Examples of the first group are (a) Helio Red (Toluidine Red)--D. & C. Red No. 35 m-nitro-p-toluidine +/•-naphthol (b) Hansa Yellow--Ext. D. & C. Yellow No. $ m-nitro-p-toluidine + acetoacetanilide. Examples of the second group are (a) Lake Red C -- D. & C. Red No. 5 C acid + /3-naphthol (b) Lithol Rubine 4B -- D. & C. Red No. 6 4B acid +/g-oxynaphthoic acid. These two pigments are readily converted to the toners by boiling under suitable conditions with a soluble heavy metal salt as illustrated: Sodium Lake Red C + BaCL -• barium toner of Lake Red C, com- monly known as Bronze Scarlet -- D. & C. Red No. 9. Sodium Lithol Rubine + CaCi• -+ calcium toner of Lithol Rubine, commonly known as Rubine Toner 4B - D. & C. Red No. 7. It should be appreciated that complications can easily creep into the nomenclature of these toners. In the casc of Lithol Rubine, it is possible to
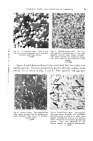
6 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS prepare the sodium, strontium and barium salts. These toners can all be resinated with 5-50 per cent of resin giving a broad range of shades, all of which are described as 4B toners. The term "4B toner" is therefore an indi- cation and not a precise description of the toner. Although the group of azo derivatives include most of the organic pigments there are a number which are not of this family, the most important being the phthalocyanines. These pigments are all semi-coordinated compounds containing a metal as the central atom in the linkage system. The metal may be copper, nickel, etc., each giving a variation in the final shade. For this reason their use in cosmetics is not widespread. Their special properties nevertheless render them most useful in certain applications. Vat colours have been mentioned in connection with the production of lakes and colours of this type are sometimes employed as pigments on their own. This again occurs only in certain specialised situations where the normal azo pigment fails to be satisfactory. PHYSICAL AND CHEMICAL PROPERTIES, TESTS AND IDENTIFICATION OF LAKES AND PIGMENTS It should be realised that no group of these materials either as set out previously or if put into chemical divisions, gives the same chemical and physical properties as a group. Each pigment or lake must be considered as a separate entity and, although each group may give a certain property to all of its members, this will vary between each of them in accordance with the particular method of manufacture. As an illustration of this, one might take a basic dyestuff such as Brilliant Green. If converted into a lake, using oleic acid as a precipitating agent, a bright but light-fugitive and heavy-bleed-in-oil lake is obtained. When using tannic instead of oleic acid the lake is dull in shade, of slightly increased fastness to light and reasonably non-bleeding in oil. When using a complex acid such as phosphomolybdotungstic acid, one obtains a lake very bright in shade, fast to light and reasonably fast to oil. With fastness to light, the amount of associated materials present has a considerable effect on the behaviour of the lake or pigment. It is well known in paint testing that the fastness to light of the paint lies in the pigmentation and not in the pigment itself, and a coal tar pigment when exposed to light may have excellent fastness in contrast to talc, which may rapidly fade. The fastness to light of the pigment may be given by the manufacturer as a comparison with the Madder scale (1-10) or the Wool scale, but is more often given as excellent, good, fair or poor. This is only an indication of this par- ticular property and often refers to the exposure of the full-strength pigment. Further tests are usually necessary and accelerated tests using a carbon arc
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)