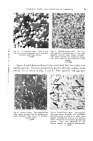
PIGMENTS, LAKES AND DYESTUFFS IN COSMETICS 17 be made up from the listed name of the colour from which they are prepared, from the name of the radicle combined with this colour together with the word "Lake." It should be noted that apart from the minor exception of the external colouring of shell eggs, lakes are not permitted under the F.D. & C. Act for the colouring of food. Hence any lake prepared from an F.D. & C. colour is automatically demoted to the D. & C. section, e.g. a lake prepared by extending the aluminium salt of F.D. & C. Yellow No. 5 (Tar- trazine) upon a substratum is called D. & C. Yellow No. 5--Aluminium Lake, while the lake prepared by extending D. & C. Red No. 9 (Lake Red C-- Barium toner) upon a substratum is D. & C. Red No. 9--Barium Lake. The Act also introduced the process of certification of these coal-tar colours under which the colour manufacturer has to forward for analysis a large sample of the colour taken from the manufactured bulk to the Food & Drug Administration in Washington. If approved, the batch of colour in question is given a lot number and every sale of this batch must quote this reference with the manufacturer's batch number and the percentage purity of the dye. It will be appreciated from the above condensed details that the use of colours in cosmetics in the United States of America is strictly controlled and all who have had dealings with the provisions of this Act will pay tribute to the foresight and meticulous care and attention which has been given to this matter by our American friends. One must remember that this Act was introduced in 1938 (20 years ago) and although minor alterations have been made since its inception the basic principles of the U.S.A. Food, Drug & Cosmetic Act are the guiding light in all present-day work on the controversial subject of "chemical additives" to food and cosmetics. There are now 12 F.D. & C. colours in the first Section, all of which are soluble dyestuffs (1 violet, 2 blues, 3 greens, 2 yellows and 4 reds) and four additional dyestuffs are at present the subject of investiga- tion. In the D. & C. Section, there are now 68 colours (16 of which are pig- mentary and the remainder water or oil-soluble), while in the Ext. D. & C. Section there are 33 colours (37, if the 4 F.D. & C. colours under suspicion are included), of which 2 are pigmentary and the remainder soluble. The numbers quoted above for each Section do not include lakes which can be prepared from the permitted dyestuffs. It is of interest to note that two colours on the Schedule are prepared by processes which are patented by a cosmetic manufacturer and which can presumably not be used by others. When a colour comes under the suspicion of being harmful and this is proven, then the Food & Drug Administration usually de-classify the colour to the last section, i.e. an F.D. & C. is reduced to an Ext. D. & C. (F.D. & C. Orange No. 1 is now Ext. D. & C. Orange No. 3).
18 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS C 6t •t6td6t . The proximity of the two countries from a geographical viewpoint tends to ensure that their legislation on non-political matters runs on parallel lines and the Canadian Food & Drugs Act is in many aspects similar to the U.S.A. Food, Drug & Cosmetic Act. With foodstuff colours, there is a list of permitted dyestuffs with specifications but, as distinct from the U.S.A., no certification procedure with analysis of samples. With cosmetics Canadian law calls for the use of colours with specified limits for As, Pb and heavy metals (1.4 as against 2.0 ppm for As, 20 ppm for Pb for both countries and 100 ppm as against 30 ppm for heavy metals). There is, however, no list of permitted colours for cosmetics, the authori- ties being content with the regulation that adequate samples of any colour used in a cosmetic shall be furnished to them on written request. This, no doubt, ensures that the manufacturer uses an approved colour and, in conjunction with the fact that many producers in the U.S.A. have subsidiary or associated plants in Canada, the colours employed in that country are essentially based on the U.S.A. schedule. United Kingdom. As yet, there is no permitted or approved list of colours for cosmetics and beyond the very general provisions of the Food & Drugs Act manufac- turers may use any colour at their discretion. The same position applied until very recently to foodstuff colours where a comparison with the U.S. system is of interest. The first regulations governing the use of colours in foodstuffs were issued in 1925 and represent a distinct departure from the United States principle of permitted colours. These regulations specified that five coal-tar colours were prohibited for use in foods and that, apart from certain wide provisions in the Food & Drugs Act, any other colour could be employed for this purpose. This state of affairs continued until 1947 when a Food Standards Committee was appointed by the Minister concerned, to review the current regulations under the various Acts and Statutory Orders governing the composition of foods and, in turn, a Preservatives Sub-Com- mittee was set up one of whose interests was foodstuff colours. In 1954 this Committee published a report on colouring matters which included several recommendations designed to revise radically the current legislation. The concept of "permitted colours" was introduced, in the same way as in the U.S.A. Food, Drug & Cosmetic Act and the Sub-Committee stated that they had almost adopted the U.S.A. list of food colours as such. In the light of the present position, it is perhaps a great pity that they did not adhere to this decision. A list of recommended permitted colours, the "tentative list," was published which detailed 32 colours described by their scientific nomen- clature. Specifications were not, however, given for the individual colour. The matter was then fully discussed with the various industries concerned
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)