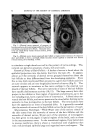
RHEOLOGICAL MEASUREMENTS IN THE COSMETIC INDUSTRY 95 Recently, there has been a description of an instrument for measuring spreadability. It is described as a "grindometer" and used in the printing industry. •a(zek (10) suggests it can be applied to measuring this property of cosmetic preparations. Obviously for cosmetic purposes the Figure 2.--Extrudiometer. The apparatus consists of a mercury head in the form of a U-tube, the right-hand leg being graduated in cc. Air is forced into the right-hand leg .through a two-way tap so that the air pressure is a few centimeters of mercury greater than a predesigned standard. By switching the tap over, the air under pressure can be passed into the tube of paste and held in a jacketed tube which is kept at a temperature of 20øC., by circulating water from the thermostat. The tube of tooth paste is fitted with a standard cap and nozzle. As the compressed air is introduced, the paste is expressed and when the pressure has fallen to a standard height of mercury, the time taken for the level of the mercury to rise by 5 cc., is determined. This time is a practical measure of the extrudability of the paste. determination should be car- ried out at some temperature below 37øC. The instrument consists of a sliding plane which moves over a small trough which is inclined at an angle of 170 ø to the sliding plane. A small measured quan- tity of the material is placed at the deep end of the trough and the slide bar slowly moved along so that the preparation then is spread. By measuring the length of the spread, values can be obtained which, if ex- amined statistically, can indi- cate whether modification of the formula increases or not the ease of spreading. I have no experience whatsoever of this apparatus but it would seem to be one which might well be considered as a means of giving a value to the ease of spreading of cosmetic prepara- tions. EXTRUDABILITY Another aspect of viscosity which is important concerns paste-like preparations which have to be extruded from a tube. The best example, of course, is tooth paste. Here the ease with which the contents of the tube can be squeezed on to the bristles of a tooth brush is of great importance and, indeed, it can be said that small changes in extrudability are easily noted by the consumer. Here the problem is one which includes many theological properties of which thixotropy and rigidity are important. An apparatus of suitable type was
96 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS described by de Wade in his paper (loc. cit.). Obviously the best test is not by means of some discrete instrument but to use a tube of tooth paste as the container. A simplified version was devised by my colleague, }'ugh, and it has been found to function in a very satisfactory fashion (Fig. 2). The tube of tooth paste is stored at constant temperature, 20øC., and the crimp of the tube is opened, carefully rounded and a rubber bung with a glass tube passing through it, is firmly inserted. The tube is held in a jacketed holder through which water at 20øC., is circulated. The cap of the tube is replaced by a cap fitted with an open orifice of precise dimen- sions and the outlet just protrudes from the bottom of the jacketed holder, the tooth paste tube being retained by the shoulder. By means of a head of mercury, air is forced into the tube and after allowing a few ccs. to be forced out so that an initial standard pressure is reached, the time taken for five mls. of the paste to be extruded is measured. This time is, therefore, in- versely proportional to the property which is called extrudability. No at- tempt is made to transpose the time of extrusion into C.G.S. units for a number of reasons, one being that the pressure is not constant throughout the determination it is simply not necessary. One interesting feature of theology which can be well brought to notice in the case of tooth pastes deserves some consideration. As you all know, tooth paste is essentially a dispersion of a solid in a liquid which has been made viscous by means of protective colloids and inevitably the system re- quires a surface-active material, as for example, soap or synthetic detergent. It is well appreciated that when a powder is dispersed in a liquid, provided the particles are in suspension, the viscosity is increased. This apparent viscosity was investigated by Einstein who showed that, at infinite dilu- tion, its value could be calculated by means of the equation: t/c = t/(1 d- 2.5c) when c = volume of spheres Work by Kynch (11) has shown that this equation holds only for concentra- tions of less than 1 per cent. This sort of thing holds for many properties of matter and far too often physical-chemical equations which relate basic- ally to dilute solutions are murdered in attempts to apply them in practice where the conditions are beyond their applicability. Thus, whilst the effect of a solid will vary at high dilution according to t. he physical-chemical properties of the substance itself and, indeed, of the medium in which it is dispersed, nevertheless, when it comes to pastes, other factors have to be considered. In order to look at this problem, it is a good plan to consider what is known of the interfacial boundary between two forms of matter. A bound- ary between a liquid and solid or, indeed, liquid and a gas, is not just a molecular film. It is of some significant thickness although continually changing as the distance between the layers and the surface increases. If, therefore, one has a high concentration of solids in a tooth paste then the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)