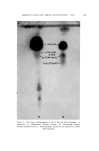
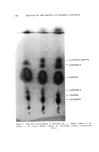
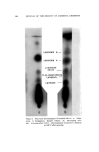
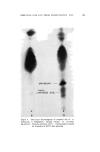
J. Soc. Cosmetic Chemists, 16, 119-131(1965) Approaches to a Quantitative Method for Testing Eye Irritation SaM P. B/txxISXa, M.S., and EuC,•N• S. McSw•N•¾, JR., A.B.* Presented September 24-25, 1963, Seminar, Boston, Mass. Synopsis--The validity of the Draize eye irritancy test has been recently questioned, and its use has become the subject of considerable controversy. An experimental approach used to determine the irritancy of industrial materials in our laboratory has been found to provide quantitative and reproducible data. This method is based upon the response obtained in the eye as a function of different exposure times. These exposures are varied in an attempt to ascertain the maximum level of exposure which the tissue will tolerate before ocular changes are produced. Two or more additional points are included at longer exposure times in an attempt to elicit discernible changes. The eyes are examined, and the lesions scored. From the data, a time-response curve is constructed for each sample. It is possible to compare the relative irritancy of different samples by estimating the exposure time required to produce equivalent lesions. Both rabbit eyes are instilled either with two different test materials or with a test material and a standard. The use of a new corneal eye applicator has greatly im- proved the reproducibility of results obtained with viscous materials. I. INTRODUCTION Within the last five years, the American public has become much more safety conscious with regard to the use of drugs, cosmetics, and household products. Public awareness is usually translated into new or supplemental regulatory legislation and in some instances leads to more stringent enforcement of the existing regulations. In the case of household products, Congress enacted the "Federal Hazardous Sub- stances Labeling Act" (July 12, 1960). Included in the regulations pur- suant to the provisions of the act are methods for testing and criteria for establishing precautionary labeling in cases of hazardous materials. No such legislation is required for cosmetics, since the requirement for * Arthur D. Little, Inc., Life Sciences Div., Cambridge, Mass. 119
120 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS safety of these products is already covered in the Food, Drug and Cosmetic Act. Interpretive regulations for cosmetics have not been issued. Unless we can accurately assess the hazard of a product, however, the mere enactment of such a law obviously does not constitute or ensure consumer protection. Methods of testing and, more important, the criteria for evaluating the hazards of a product have in many cases not been clearly defined. In addition, as long as animals and not humans are used as the principal subjects for establishing the degree of product hazard, there is always a risk of marketing a product which appears safe when tested on animals but is injurious to man under certain conditions. Although the choice of methods to be used is left to the investigator, it is usually more expedient to use officially accepted tests in situations where government regulations must be followed. Within the last several years, however, various groups have seriously questioned the rdiability of one of these tests, namely, that used for eye irritants (1). The Draize test (2, 3) is used almost exclusively in this country for testing eye irritants, although other methods have been reported. Friedenwald (4), in his studies on acid-base tolerance, employed a drip method (1 every 10 minutes) for instilling samples into the eye in an attempt to control the level of exposure. He gave individual scores for the degree and extent of corneal opacity, iritis and conjunctivitis and then added them to obtain a total index of irritation. Carpenter and Smyth (,5) carried out a very extensive eye testing program which in- cluded a large number of chemicals. In addition to using a point scoring system to describe the degree of corneal opacity, iritis and keratoconus, these investigators also included a score for the degree of necrosis seen after staining the eye with fluorescein. Kay and Calandra (6) recently devised a system of classifying ocular lesions ob- tained by the same exposure procedure as outlined by Draize, except that they based their scoring system on the extent of irritation, its per- sistence, and the over-all consistency of the data. Test materials are given tentative ratings on the basis of the scores obtained from all three tissues (cornea, iris and conjunctivitis) within the first ninety-six hours. These tentative ratings are adjusted according to the mean total scores at various specified times. The first point to be considered with the Draize test, or any of the .other procedures for that matter, is the ultimate objective. Most of those who use the test agree that it is intended to provide a measure of the irritancy of a product by exposing animals (rabbit) under the con-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)