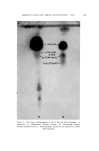
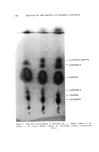
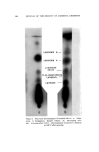
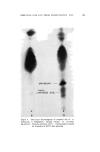
J. Soc. Cosmetic Chemists, 16, 169-184(1965) Water-Insoluble Bacteriostats Solubilized in Soap and Detergent Solutions KENNETH L. RUSSELL, Ph.D.,* and STEPHEN G. HOCHt Presented before the Third Congress of the I.F.S.C.C., June 21-26, New York City Synopsis--Three nonionic surfactants, ethoxylated lanolin alcohols, ethoxylated sorbitan esters and ethoxylated nonyl phenol, were used to solubilize two water-insoluble bacterio- stats, trichlorocarbanilide and mixed di- and tribromosalicylanilide, in four surfactant solu- tions. The maximum percentage of each bacteriostat solubilized under specific conditions by each nonionic in 20% potassium coconut soap, 20% triethanolamine lauryl sulfate, 20% triethanolamine dodecyl benzene sulfonate and a mixed amphoteric-anionic solution is re- ported. Only the ethoxylated lanolin alcohols were found to permit simultaneous solubiliza- tion of the bacteriostats and 1.0% lanolin oil. The inclusion of the latter increased the amounts of the bacteriostats which could be solubilized in two of the detergent solutions. The bacteriostatic and bactericidal properties of the two antibacterials solubilized in tri- ethanolamine lauryl sulfate are compared with those of the same agents suspended in the surfactant without the nonionic solubilizer. It was found that the bacteriological activity of the solubilized agents was as great as or greater than that of the suspended materials. Three previous papers by the authors (1-3) have reported on studies of the solubilization of lanolin oil in detergent solutions and on properties of the resulting clear aqueous products. The solubilization of the water-insoluble oil in an aqueous system containing mainly an anionic surfactant was accomplished by heating the oil with a relatively small proportion of a nonionic material and adding this mixture to the heated anionic surfactant solution. The nonionics employed were ethoxylated lanolin alcohols or diethanolamides of coconut fatty acids * Consultant, 245 Grant Ave., Nutley, N.J. 07110. t Maimstrom Chemical Corp., Linden, N.J. 07036. 169
170 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS or combinations of these two materials. The mild emollient detergent products so prepared have been shown to possess properties making them highly suitable for cosmetic shampoos and skin cleansing preparations. During the past several years a trend to the use of antibacterial and antifungal agents in shampoos and skin cleansing products has been evidenced by the growing numbers of anti-dandruff shampoos, anti- bacterial liquid cleansers and antibacterial soap and detergent bars. These products, generally classified as drugs, often employ antibacterial agents which are poorly soluble in water. Some of the agents used are only slightly water soluble but may be fairly readily solubilized in significant concentrations in soap or anionic synthetic surfactant solu- tions, usually with the aid of heat. Examples of compounds of this type are 2,2 '-methylenebis [3,4,6-trichlorophenol ] (hexachlorophene, G- 11 ©*) and bis(2-hydroxy-5-chlorophenyl) sulfide (S-71'). Some other antibacterials in use are not so easily solubilized and re- quire added chemicals and careful techniques to solubilize them in significant amounts, even in soap or other anionic surfactant solutions. Examples of these practically water-insoluble antibacterial agents are 3,4,4'-trichlorocarbanilide (TCC•) and the mixture of 5,4'-dibro- mosalicylanilide and 3,5,4'-tribomosalicyanilide marketed under the name of Diaphene.õ The use of this type of bacteriostat is usually limited to opaque products where the material is suspended and remains substantially in its insoluble state. It was felt that a study of the solubilization of this latter type of bacteriostat in aqueous solutions of soap and several synthetic detergents would be of general interest. Another purpose of the study was to determine the effects, if any, of simultaneous solubilization of lanolin oil and the bacteriostats in the surfactant solutions. The present paper describes the method of solubilization employed using three polyoxeth- ylene nonionics. It also reports on the results of some bacteriological testing of selected samples of the products containing the solubilized materials. SOLUBILIZATION Recognizing at the start of the study that the approach would have to be by a "trial-and-error" method, certain materials were selected and * Sindar Corporation. t Norda Essential Oil and Chemical Co. • Monsanto Chemical Co. õ Trade Mark of Stecker Chemicals Inc
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)