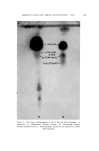
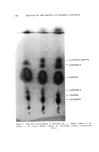
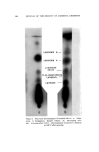
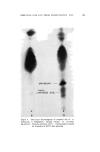
J. Soc. Cosmetic Chemists, 16, 155-168(1965) Essential Oils and Their Constituents. XXV* © Thin Layer Chromatography. Some Chemical and Chemotaxonomic Applications M. C. NIGAM, Ph.D., t I. C. NIGAM Ph.D., A.R.I.C., LEO LEVI, Ph.D., F.C.I.C.:• Presented before the Third Congress of the I.F.S.C.C., June 21-26, New York City Synopsis--Thin layer chromatography has been applied to the analysis of essential oils and their constituents. Rf-data are given for terpene alcohols, esters and ketones. Vanillin- sulfuric acid (5% w./v.) is used as spray reagent and sensitivity limits are reported for the compounds examined. Characteristic colorations observed are also recorded. Typical thin layer chromatograms are shown to illustrate the detection of sophistication of a com- mercial bois de rose oil, the distinction between genuine palmarosa and gingergrass oils, and the recognition of botanical and/or geographical origins o[ authenticated spearmint and citronella oils. INTRODUCTION Within a short period of time thin layer chromatography has be- come a most important technique for the identification, characteriza- tion and determination of chemical compounds as well as complex mix- tures. In the authors' laboratory the technique has been used with * Part XXIV, Journal of Chromatography, in press. t National Research Council o[ Canada Postdoctoral Fellow, 1963. ** Pharmaceutical Chemistry Division, Food & Drug Directorate, Ottawa, Ont., Canada. 155
150 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS considerable success for the analysis of essential oils and their constit uents. It is the purpose of this paper to report some specific chemical and chemotaxonomic applications of interest to the cosmetic industry. EXPERIMENTAL Apparatus Thin Layer Chromatographic Assembly: Desaga Unit.* , According to Stahl C. Materials Adsorbent: Silica Gel G (Merck). Spray Reagent: Vanillin in concentrated sulfuric acid, 5% w./v. Essential Oils and Essential Oil Constituents: For sources of supply see acknowledgment section. Table I Thin Layer Chromatography of Alcohols Compounds Sensi- •- --Color Observed --• Relative tivity Room After Rf •, Temperature Heating Value • Limonone diol 1 Faint pink Deep pink Geraniol 5 Grayish purple Faint violet trans-p-Menthadiene-2,8-ol-1 5 Faint blue Blue black Nerol 5 Bluish green Violet Citronellol 1 Faint violet Violet trans-Carveol 1 Violet Violet Dihydro Carveol 1 Faint violet Bluish violet Isopiperitenol 5 Faint blue Deep blue Perillyl Alcohol 5 Faint blue Deep blue a-Terpineol 1 Faint violet Violet cis-p-Menthadiene-l(7),8-ol-2 5 Faint blue Blue black cis-Carveol 1 Violet Violet Menthol 10 ... Faint violet •-Terpineol 1 Faint violet Violet Linalool 1 Faint violet Violet Borncol 20 ... Faint grey Isopulegol 1 Faint brown Violet /%Fenchyl Alcohol 5 Orange Reddish brown Carvomenthol 100 Faint green Deep green Neomenthol 10 Faint grey Faint violet a-Fenchyl Alcohol 100 . .. Faint violet 0.31 O. 72 O. 80 O. 82 O. 86 0.95 O. 98 O. 98 O. 98 1.00 1. O0 1.01 1.02 i 04 i 06 i 07 1 12 i 25 i 26 1.28 1.30 • Reference Standard: a-Terpineol. Developing Solvent: Benzene-Methanol, (10:1). * Manufactured by Desaga, Heidelberg, Germany. U.S. Agent: Brinkman Instruments Inc., Cantiagne Rd., Westbury, N.Y.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)