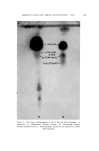
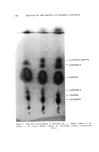
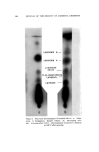
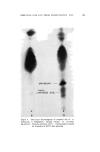
SOLUBILIZATION OF BACTERIOSTATS 183 The bactericidal action is not sufficient to be measured by a short time killing test, such as the phenol coefficient test. Bactericidal action is apparent, however, at fairly low active ingredient concentrations in a longer term test, as shown in Table VI. Bacteriostatic action against S•aph. aureus, by both test methods used, is apparent at lower concentrations of TCC than of Diaphene or G-11. No one of the three antibacterials tested shows bacteriostatic action against E. coli at sample dilutions of 1-125. Taking all the data into consideration, the presence of the nonionics as added solubilizers does not appear to reduce the antibacterial action of the active ingredients. In the experiments where the samples without nonionics (suspensions) were shaken throughout the twenty-four-hour incubation period, with the intention of obtaining maximum action from the insoluble active ingredients, the antibacterial action turned out to be less than that from the solubilized samples. The presence of solubilized lanolin oil in the systems appears to have little, if any, effect upon the antibacterial activity. It appears neither to interfere with nor enhance the activity of the active agents. The apparent greater activity of sample H-3/6 in which TCC was solubilized by Igepal CO-630, in comparison with the solubilization by Nimcolan S or Tween 80, should be checked. If confirmed by repeti- tion, this combination would seem worthy of further investigation. Substantivity on fresh calf skin of the antibacterial agents was as good from the samples in which the agents were solubilized as from those in which they were suspended. Likewise, the persistence or loss of activity through aging of the treated calf skin samples at high relative humidity was not dependent upon treatment with solubilized or sus- pended antibacterial agent. (Received June 26, 196•) REFERENCES (1) Russell, K. L., and Hoch, S. G., Drug and Cosmetic Industry, 90,294 (1962). (2) Idem., Proc. Sci. Sect. Toilet Goods Assoc., 3?, 27 (1962). (3) Idem., Ibid., 38, 51 (1962). (4) McBain, M. E. L., and Hutchinson, E., Solubilization, Academic Press, New York, N.Y., 1955, pp. 199-203. (5) Charles, R. D., and Carter, P. J., J. Soc. Cosmetic Chemists, 10,383 (1959). (6) Methods of Analysis of the Association of O.t•cial Agricultural Chemists, 9th Ed., Asso- ciation of Official Agricultural Chemists, Washington 4, D.C., pp. 63-65. (7) Walter, G. R., and Gump, W. S., J. Soc. Cosmetic Chemists, 13,477 (1962).
184 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS (8) Sindar Pamphlet "Antimicrobial Activity of G-11 and Competitive Products." (9) Roman, D. P., Barnett, E. H., and Balske, R. J., Proc. Sci. Sect. Toilet Goods Assoc., 28, 12 (1957). (10) Stecker, H. C., and Faust, R. E., J. Soc. Cosmetic Chemists, 11,347 (1960).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)