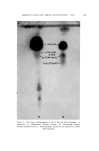
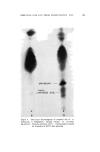
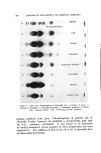
ESSENTIAL OILS AND THEIR CONSTITUENTS. XXV. 157 A nalytical Procedures Chromatoplates were prepared by spreading a slurry of Silica Gel G in distilled water (1:2) uniformly over clean glass plates (20 cm.2). The layer (thickness 250/•) was allowed to dry for fifteen to twenty min- utes and activated by heating in an oven at 120øC for one hour. The plates were then placed into a specially designed chamber (1) and used after storage for twenty-four hours. Following the application of analytical samples, they were developed by means of suitable solvent systems and sprayed with the reagent for color development. In some instances they were heated for about five to ten minutes at 105øC to observe additional characteristic colorations. Tentative identification of spots was achieved by comparison of l•f-values with those of authentic reference standards. Confirma- tion was obtained by infrared and gas chromatographic examination of constituents isolated by extraction of adsorbent material from the ap- propriate area of the chromatoplate. RESULTS AND DISCUSSIGN Thin Layer Chromatography of Terpene Alcohols, Esters, A ldehydes and Ketones The different polar characteristics of these classes of compounds re- quire the use of different developing systems. Benzene was found to be a suitable solvent for esters, while a mixture of n-hexane and ether (20:3) was applied to resolve carbonyl compounds. Alcohols were de- veloped with a benzene-methanol solution (10: 1). Several of the ter- penoids examined displayed characteristic colorations when sprayed with the reagent, an observation which aided materially in the identifi- cation of compounds exhibiting similar migration properties. Heating the chromatoplate after spraying was found to bring about further characteristic color changes and increased moreover the sensitivity of the reagent, making it suitable for the detection of trace constituents. Tables I, II and III record the experimental observations. Analysis of Essential Oils Preliminary experiments to determine the nature of constituents present were carried out, using a mixture of n-hexane-ether (20:3) as developing solvent. The observations made served to select and apply a more suitable system to achieve the desired resolution.
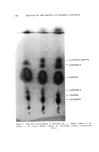
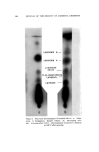
158 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table II Thin Layer Chromatography of Esters Sensitivity Compound T Color Observed Relative Rf Value s Geranyl Acetate 2 Faint blue Linalyl Acetate 2 Faint blue Isobornyl Acetate 10 Orange trans-Carveyl Acetate 2 Violet blue Terpinyl Acetate 2 Faint blue Bornyl Acetate 20 Faint purple cis-Carveyl Acetate 2 Violet blue Linalyl Formate 2 Faint blue Menthyl Acetate 50 Violet blue •' Geranyl Formate 2 Faint blue Menthyl Isovalerate 20 Deep blue b 1.00 1.13 1.14 1.17 1.19 1.23 1.28 1.41 1.40 1.59 2.08 Reference standard: Geranyl acetate. Developing solvent: Benzene. On heating. Table III Thin Layer Chromatography of Carbonyl Compounds Compounds Sensi- .-- -Color Observed-----• tivity Room After 3' Temperature Heating Relative Ri Value s Piperitenone 1 Faint purple Faint blue Piperitone 100 ... Saffron yellow fi-Ionone 1 Faint blue Faint blue Citral 10 Violet Violet 3'-Ionone 1 Faint blue Faint blue Dihydrocitral 200 Violet blue Violet blue Carvone 1 ... Faint violet Pulegone 10 Faint pink Purple a-Ionone 1 Faint blue Faint blue Carvomenthone 100 ... Faint pink Methyl Heptenone 1 Faint violet Green Menthone 1000 ... Faint brown Dihydrocarvone 200 Faint pink Purple Citronellal 1 ... Faint violet Camphor 1000 Faint blue Faint blue 0.75 0.77 O. 78 O. 82 0.92 O. 94 1. O0 1. O0 1. O0 1.01 1.02 1.17 1.36 1.47 1.72 a Reference Standard: Carvone. Developing Solvent: n-Hexane-Ether (20:3).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)