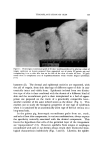
148 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 4000 30•00 2000 1500 CM" 1000 900 800 700 ,oo ................... t:•' .7,:',',•T:•.:'•7'•" "J ...... '=•' •. '-:¾ :' ..... '. i•- .•: ,oo z 6C , • .... , •j ,/ m m• • ' . •. i• , • ' :' ' • j LJ • •.. •..• •:•, 60 '• •i• • • -•l• • • t •':ti, H!HIIh',4LZ 1,2• J• ,"I.Z I •[•!•_/ •i' 20 •'r.•ffl•,..•-•,•lq•h•t•'t-J!fttlt•IP• , ,•.•iiT•-l,• l 3 4 5 6 7 8 9 10 II 12 13 14 WAVELENGTH (MICRO•) Figure 6. Crude 2-amino-5-nitrophenol 4000 3•00 2000 1500 CM'• 1000 900 800 700 IOO 8c 8o •6C 60 i.- •40 40 3 4 5 6 7 8 9 10 I1 12 13 14 15 WAVELENGTH (MICRONS) Figure 7. Purified 2-amino-5-nitrophenol In the laboratory recrystallization of 2-amino-5-nitrophenol from water a dark brown material was removed during the hot filtration. This material was washed repeatedly, on the filter, with hot water, until the flitrate came through colorless. After drying at 50øC this •oTay-brown powder melted at 220øC. It had an ash content of 2.36% and an iron content of 1.36% determined and reported as ferric oxide. An infrared spectrogram of this sludge is shown in Fig. 8. 40003•00 2000 1500 CM-' 1000 900 800 700 100 •4c ' t,• Jl '.'i ' 3 4 5 6 7 8 9 10 II 12 13 14 150 WAVELENGTH (MICRONS) Figure 8. Sludge h'om 2-amino-5-nitrophenol
INTERMEDIATES FOR HAIR COLORS 149 The infrared spectrogram of the sludge from 2-amino-5-nitrophenol differs in so many ways from that of the pure or crude product that there can be no doubt that it is an entirely different compound. However, insufficient data are available to warrant any speculation as to its struc- ture. The amount of material precipitated in the boiling experiment, pre- viously described, was 0.10% for the crude and 0.02% for the purified 2-amino-5-nitrophenol. 2-A mino-4-nitrophenol 2-Amino-4-nitrophenol can be prepared by the reduction of 2,4-dini- trophenol with sodium sulfide, followed by neutralization and crystalliza- tion from water (11). Its solubility at 25øC in aqueous solution at a pH of 9.7 is $.47% (1). Comparable analyses of this intermediate before and after recrystallization, as a production lot, are given in Table V. Unlike the other nitro intermediates considered in this paper, this compound forms a hydrate. The water can be removed by a prolonged drying at 65øC. Table V Analyses of 2-Amino-4-nitrophenol Purified Raw Purity, % 97.9 75.0 Ash, % 0.07 0.06 Iron, ppm 69 230 Melting point (Fisher-Johns), øC 145.7 . . . Melting point of hydrate, øC 110.0 93.0 Water, % . . . 11.6 In the ageing tests, precipitation of the solution containing the crude product started after 15 days both at 50øC and at room temperature, while no precipitate appeared in bottles containing the purified material after 4 weeks. The infrared spectrograms of the crude and purified 2-amino-4-nitrophenol showed no significant differences. During the recrystallization of 2-amino-4-nitrophenol from water, it .gave off an odor similar to that of exploded firecrackers. The infrared spectrogram of this sludge showed no relationship to those of the crude or purified products. It was found to be insoluble in all the organic sol- vents tested and no significant bands appeared in the in[Tared spectro- gTam, indicating that it is most likely a product formed by decomposi-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)