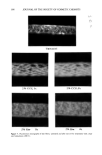
DETERMINATION OF ASCORBYL DIPALMITATE 363 Quantitation of ADP is accomplished by use of the DuPont Advanced DSC Data Anal- ysis Program, which is used to integrate the endotherm that occurs between 56 ø and 58øC. The energy that is absorbed during this transition is integrated and displayed in Joules per gram of sample. The weight percent ADP can be calculated manually by a ratio of the endotherm of the sample to the endotherm of a pure standard of ADP according to the following [c = ascorbyl dipalmitate in the sample (%) U = sample endotherm (Joules/g) and S = ascorbyl dipalmitate standard endotherm (Joules/g)]: 100 U C -- The enthalpy in Joules/g of pure ADP of the endotherm between 56 ø and 58øC was determined to be 30.4 Joules/g (RSD = ---0.7 N = 3). Alternately, a calibration curve of enthalpy versus percent ADP in an inert medium such as talc can also be determined and plotted. The percent ADP in an unknown whitening powder can then be determined directly from the graph (Figure 2). Figures 3 a-d show the thermograms of whitening powders produced by four cosmetic companies. It is interesting to note that in these commercially available mixtures, the endotherm that corresponds to the melting point of ADP is lowered to approximately E 3O 25 2O 15 10 ! ! i i ! , ! t 10 20 30 40 50 60 70 80 90 100 Weight percent ADP in talc Figure 2. Plot of enthalpy versus weight percent ADP in talc.
364 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS ! I ! i I i i i ! i i ! 20 30 40 50 60 70 80 90 100 110 120 130 Temperature (C) Figure3a I i i i i i i • 20 30 40 50 60 70 80 90 100 110 120 130 Temperature (C) Figure3b Figure 3. DSC thermograms of commercially available whitening powders: 3a: Company A. 3b: Company B. 3c: Company C. 3d: Company D.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)