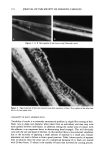
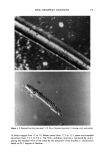
156 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table IIIb Effect of Increasing Palmitic Acid on Efficiency of Cholesterol and Palmitic Acid Incorporation Into Liposomal Preparations (cholesterol sulfate maintained at 25%) % Added Formu- Choles- % Added palmitic % Lipo- lation Cera- Choles- Palmitic terol cholesterol acid Trapped somes number mides terol acid sulfate incorporated incorporated volume formed? 11 46.1 28.9 0.0 25.0 54.0 (4.2) 1.6 (0.2) yes 6 43.1 26.9 5.00 25.0 83.8 (2.9) 83.7 (2.6) 2.1 (0.3) yes 7 40.0 25.0 10.0 25.0 80.9 (1.8) 84.2 (2.7) 2.6 (0.2) yes 8 33.9 21.1 20.0 25.0 81.9 (5.3) 76.0 (3.5) 4.0 (0.2) yes 9 15.4 9.6 50.0 25.0 81.9 (6.6) 55.9 (3.5) 0.9 (0.0) yes 10 0.0 0.0 75.0 25.0 -- 25.6 (0.4) 2.1 (0.3) yes 12 0.0 0.0 100 0.0 -- 4.3 (0.9) 1.0 no All liposomes were prepared by the conventional hydration method. All values expressed as weight percent. Results expressed as average values (standard deviation). lipid remnants seen on top of the column and the virtual absence of liposomes in the void volume. Stratum corneum lipids did not form liposomes readily until the hydration temperature was raised to 80øC, where phase separation was apparent. Thin-layer chromatography of the stratum corneum lipid extract showed spots that corresponded to ceramides, pal- mitic acid, cholesterol, and two other unidentified spots. Cholesterol sulfate could not be detected, perhaps due to the low detection limits. Addition of 10% cholesterol sulfate to this mixture, however, resulted in an increase in the proportion of lipid incorporated in the final liposomes (Table IIIc). The effect of temperature on the quantities of lipid incorporated into liposome formulation 2 is shown in Table IV. Phase separation was evident when this lipid composition was hydrated at 80øC. Unlike other lipids used in the experiments, cholesterol sulfate is a bilayer former. Lipsomes prepared from cholesterol sulfate alone are large and exhibit a high trapped Table Illc Demonstration of the Inability to Form Liposomes in the Absence of Cholesterol Sulfate % Added Formu- Choles- % Added palmitic % Lipo- lation Cera- Choles- Palmitic terol cholesterol acid Trapped somes number mides terol acid sulfate incorporated incorporated volume formed? 14 55.4 34.6 10.0 0.0 0.7 (0.2) 1.0 (0.0) 13 49.2 30.8 20.0 0.0 1.5 (0.2) 1.2 (0.1) 1 46.1 28.9 25.0 0.0 2.4 (0.7) 1.9 (0.7) 15 0.0 0.0 90.0 10.0 -- 15.8 (5.4) 16 0.0 0.0 10.0 90.0 -- 66.6 (7.5) Lipid extract N/D N/D N/D N/D 36.0 (2.5) 28.2 (3.7) Extract + 10% CS N/D N/D N/D N/D 79.7 (3.9) 55.8 (3.2) 1.0 no 1.0 no 0.1 (0.0) no 0.6 (0.1) yes 2.9 (0.6) yes yes yes All liposomes were prepared by the conventional hydration method. All values expressed as weight percent. Results expressed as average values (standard deviation).
PREPARATION OF LIPOSOMES 157 Table IV Effect of Hydration Temperature on Lipid Incorporation Into Liposomal Formulation 2 Description 55 øC 80øC Cholesterol recovered in eluate Palmitic acid recovered in eluate Cholesterol in upper packing Palmitic acid in upper packing Cholesterol in lower packing Palmitic acid in lower packing Cholesterol in supernate Palmitic acid in supernate Total mass balance 84.0 +- 2.3 85.8 +- 5.7 80.5 -+ 2.8 77.3 -+ 4.2 3.3 -+ 0.2 5.9 -+ 2.0 2.6 +- 0.0 9.7 +- 3.8 4.8 -+ 0.1 4.4 +- 0.6 4.7 -+ 0.1 4.3 -+ 0.3 0.0 -+ 0.0 3.7 +- 1.7 2.2 -+ 2.0 4.6 -+ 0.4 Cholesterol --92% Palmitic acid --89% Cholesterol --96% Palmitic acid --92% All quantities are expressed as weight percent. volume. Cholesterol sulfate appears to be a vital component that renders bilayer-forming ability to the lipid mixture. Being a highly polar molecule, it may not have been well extracted by the procedure used in our laboratories, and this could be the reason why the stratum corneum lipid extract did not form liposomes readily. Addition to 10% cholesterol sulfate to the lipid extract, however, dramatically improved the bilayer- forming efficiency to the extract. The results from chromatography experiments confirm the existence of free lipids in the liposomal preparations. It raises the issue that has long been overlooked.' not all the lipids in the lipid mixture used to prepare liposomes are necessarily incorporated into the vesicles as previously assumed. For complex lipid mixtures such as those found in the stratum corneum, the final lipid composition of liposomes cannot be presumed to be the same as the compo- sition of lipid mixtures used to prepare them. Moreover, differences in the starting compositions of lipids resulted in liposomes differing in their entrapment efficiencies, final lipid compositions, and bilayer-forming capabilities. The lipid composition of the stratum corneum may, in fact, not at all reflect the lipid composition of the bilayers within the stratum corneum. There is ample evidence that a significant fraction of cholesterol (12), fatty acids (13), and perhaps other lipid components occur in separate phases within the lipid matrix of the stratum corneum. Monolayer interaction studies were carried out to determine the necessity to optimize liposomal lipid composition so that maximal bilayer-bilayer interactions can be achieved. When lipid molecules are spread on a quiescent surface, they arrange them- selves in a manner to maintain a minimum free energy. When different lipid species are combined in a ratio that favors tight packing, and when the lipid concentrations (number of lipid molecules per area) are regulated such that strong interactions are ensured, stable films are more likely to be formed. Based on this principle, the stability of the monolayers formed by various lipid mixtures was examined. A pH of 4.0 was selected as the working pH to suppress ionization of the head groups and hence min- imize lipid solubility. Therefore, a resultant unstable monolayer caused by the loss of lipid from the surface can be avoided. All individual synthetic lipids examined formed stable monolayers at pH 4. This pH was used since palmitic acid was too soluble to form a stable film at pH 7. The time required for the palmitic acid film and the cholesterol film to stabilize was inversely
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)