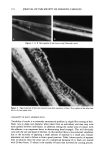
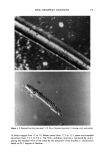
158 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS proportional to the surface pressure (-rr) of the spread film. There does not seem to be an apparent relationship between the ability of the film to form a stable monolayer and its ability to form liposomes. The trapped volumes and the surface pressures at which selected synthetic lipid mixture compositions and the stratum corneum lipid extract stabilized are tabulated in Table V. Stratum corneum lipid extract was found to form stable monolayers at a very low surface pressure (-rrstab•e = 4.4 dyne/cm), an implication of strong interactions between lipid molecules. This phenomenon is not unexpected, considering that such interaction is needed to provide high resistance to water perme- ation of the stratum corneum. Among the five original liposomal formulations examined as potential bilayer building blocks for the preparation of model membranes, the only formulation comprising all four lipid components that yielded the most stable mono- layer was formulation 2. However, large batch-to-batch variations were observed when these liposomes were prepared by the conventional hydration method at a total lipid concentration of 15 mg/ml. This was presumably due to crystallization of lipids upon removal of mixed solvent during the film formation step. Therefore, an alternative preparation procedure, reverse phase evaporation, was used. Analysis of the suspension immediately after liposome formation revealed that 94% cholesterol and 92% palmitic acid were incorporated into the vesicles. Upon 24-hr incubation at room temperature, however, needle-shaped crystals were observed. Gel permeation chromatography showed that palmitic acid content in the liposomes was reduced to approximately 35%. Mod- ifications of the working formula were then attempted, using the results from monolayer studies as guidelines. Recognizing that stratum corneum lipids formed stable mono- layers at a low surface pressure, we proceeded to optimize the composition of synthetic lipids in formulation 2 to obtain a mixture whose component lipids display high intermolecular interactions. This composition would, expectedly, lead to formation of a strong bilayer. The lipid composition was optimized to obtain a monolayer that was stable at low surface pressures. The most stable monolayer was formed when ceramides, cholesterol, palmitic acid, and cholesterol sulfate were combined using a 37.7:28.3:17: 17 weight ratio (-rrstabie = 3.8 dyne/cm). This modified formulation yielded liposomes that incorporated 93% cholesterol and 84% palmitic acid used. Table VI tabulates the changes in the lipid composition of liposomal formulation 2 before and after its com- position was modified. The modified formulation 2 (Table V) formed a stable monolayer at a surface pressure Table V Trapped Volume and •['stable of Formulations Tested by Tensiometry Formulation number Trapped volume (%) arstabie (dyne/cm) 2 3.7 2O.2 -+ O.9 2* 10.1 20.2 m 0.9 5 5.9 13.5 + 1.2 6 2.1 none 8 4.0 none 9 0.9 none 2 modified* 10.7 3.8 +-- 0.3 SC lipid extract N/D 4.4 + 0.3 All liposomes were prepared by the conventional hydration method except where noted. * Prepared by REV method.
PREPARATION OF LIPOSOMES 159 Table ¾I Effect of Modification of Formulation 2 on Efficiency of Cholesterol and Palmitic Acid Incorporation Into Liposomes % Added Choles- % Added palmitic Formulation Cera- Choles- Palmitic terol cholesterol acid % Trapped number mides terol acid sulfate incorporated incorporated volume 2 40.0 25.0 25.0 10.0 95.1 (1.8) 89.2 (4.70) 10.1 (0.14) 2 Overnight 40.0 25.0 25.0 10.0 92.8 (4.1) 31.7 (8.85) N/D 2 (modified formula) 37.7 28.3 17.0 17.0 93.3 (4.2) 85.6 (2.39) 10.0 (0.22) 2 (modified) Overnight 37.7 -28.3 17.0 17.0 95.1 (2.0) 75.1 (6.06) 10.7 (1.07) All liposomes were prepared by the reverse-phase evaporation method. of 3.8 dyne/cm, which is quite similar to the value for the stratum corneum lipid extract. This indicates that this formulation may be employed to obtain bilayers with strong interlipid interactions. In summary, these studies allowed the optimization of liposomal formulations to be used as building blocks in the formation of model mem- branes of the stratum corneum lipid compartment. The preparation of these membranes and their permeability properties using a variety of permeants will be discussed in Part II of this series. ACKNOWLEDGMENTS We wish to acknowledge Parrums Christian Dior and Liposome Technology, Inc. for support of this project. REFERENCES (1) G. M. Gray and H.J. Yardley, Lipid composition of cells isolated from pig, human, and rat epidermis, J. Lipid Res., 16, 34-44 (1975). (2) P.M. Elias, Epidermal lipids, barrier function and desquamation, J. invest. Dermatol,, 80, 445-495 (1983). (3) H. J. Yardley, "Epidermal Lipids," in Biochemistry and Physiology of the Skin, L. A. Goldsmith, Ed. (Oxford University Press, New York, 1983), pp. 363-381. (4) S. A. Long, P. W. Wertz, j. S. Strauss, and D. T. Downing, Human stratum corneum lipids and desquamation, Arch. Dem•atol. Res., 277, 284-287 (1985). (5) G. M. Gray and R. J. White, Epidermal lipid liposomes: A novel non phospholipid system, Biochem. Soc. Trans., 7, 1129-1131 (1979). (6) W. Abraham, P. W. Wertz, L. Landmann, and D. T. Downing, Preparation of liposomes from stratum corneum iipids, J. Invest. Dermatol., 87, 582-584 (1986). (7) W. Abraham and D. T. Downing, Preparation of model membranes for skin permeability studies using stratum corneum lipids, J. Invest. Demtatol., 93, 809-813 (1989). (8) E.G. Bligh and W. J. Dyer, A rapid method of total lipid extraction and purification, Can. J, Biochem. Physiol., 37, 911-917 (1959).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)