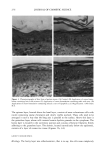
284 JOURNAL OF COSMETIC SCIENCE A •,1 A -- 17 II IIIIIII 1 I Figure 1. •0(7etting of keratin surfaces (qualitative): a. •0(7ater 00(7) in air (A). b. Oil (O) in water. c. Oil in air. Figure 2. Desorption of second layer of adsorbed cationic surfactant on keratin during rinsing. possess a mobility and/or a hydration/dehydration capacity that allows them to respond rapidly to their immediate environment.* SIMPLE CONDITIONING SYSTEMS Traditionally, cationic surfactants have been employed to "condition" fibrous systems (hair, fabrics, etc.). The early common practice was to apply the agent from relatively dilute solutions in a separate step from cleaning. The potent forces of electrostatic attraction lead to virtually immediate adsorption of a monolayer of cationic surfactant molecules on the substrate. This process, however, creates an energetically unfavorable hydrophobic surface in contact with the aqueous solution, and a second layer, with head groups "out," adsorbs on the first layer to render the surface hydrophilic (see Figure 2). During rinsing this second, less strongly bound layer will tend to desorb, leaving the primary layer as the conditioning (boundary lubricating) outer layer. Though seldom employed alone in a simple conditioning system, a high-molecular- weight polycationic agent would tend to adsorb very strongly (virtually irreversibly) on a negatively charged substrate even at very low levels of application. In both aforementioned cases the cationic agent may assist the deposition and/or spread- ing of an added oil conditioner. SIMPLE CLEANING SYSTEM A basic cleaning system (shampoo, etc.) generally consists of one or more anionic surfactant (alkyl sulfate, alkyl ethoxysulfate, etc.) employed at a 10-20% level in the base formulation. In essence, at washing concentration, the negatively charged surfactant ions are adsorbed by the soiled substrate surface, cause roll-up and removal of the soiling entities, and are themselves removed during rinsing. See Figure 3, which, for simplicity, * The presence of a covalently bonded fatty acid outer (mono) layer, whole or partial, on hair fibers will be expected to affect the details but not the essential elements of the above picture substantial hysteresis in their wetting by water is well known (2).
CLEANER/CONDITIONER SYSTEMS 285 II I I I i Iii'1 I I I ! I I Figure 3. Roll-up and removal of oil drop on keratin by anionic surfactant solutions. ignores the presence of "solid" soiling matter and the adsorption of surfactant by the keratin substrate itself. COMBINATION CLEANING/CONDITIONING SYSTEMS Although the overall performance of a combination cleaner/conditioner system for kera- tin substrates can be explained in a simple way ("removal of dirt by the main surfactant and deposition of the softening ingredient"), much of the detail and relative importance of the subprocesses involved are unknown. One can, however, attempt to list (some of) the possible events. Typically a conditioning system will comprise a soluble polymeric polycation ("1") and an insoluble oil ("2"), each alterable by mutual interaction or by interaction with the main surfactant (Table I). Here, adsorption is regarded as a mo- lecular event and deposition as a macro- or flocculation-type process. For simplicity and convenience, adsorption of the main (anionic) surfactant(s) on the keratin, which is obviously involved in the cleaning process, is omitted. While analytical information does exist on the deposition of conditioners from shampoo systems--examples are polycation (3) and silicone (4) alone and in combination--the analytical methods employed yield "net" effects measured after rinsing and/or drying, and so they provide little information about the stepwise and hence complete mecha- nisms. In particular, current explanations are incomplete regarding the influence of polycations on the deposition of silicone oil from shampoo systems (4). It is a fair but unproven assumption that the mechanism involves heteroflocculation of silicone droplets on hair fibers modified by the presence of adsorbed polycation this leads to more efficient deposition of the droplets (5). Such deposition may occur in one or both of the Table I Fate of Conditioners Washing Rinsing Drying Adsorption 1 (1 +S) Flocculation 1 + 2 (deposition) (1 + S) 2 Spreading (1 + S) 1+2 (1 + S) 2 (2 + S) 2 1: polycatiomc conditioner. 2: oil conditioner. S: surfactant (listed only in combinations with 1, 2).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)