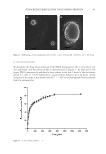
4 JOURNAL OF COSMETIC SCIENCE mechanical properties by leading to skin dryness and roughness, wrinkles, sagging, uneven skin color, and sunspots (2). In women, estrogen deficiency also contributes to aging skin. Maintaining a youthful appearance requires many cosmetic procedures and cosmeceutical products, and research is increasingly focusing on developing innovative anti-aging products derived from natural plant extracts. These products claim to offset the effects of aging however, while anti-aging products should ideally restore dermal connective tissue and epidermal components, the efficacy of most products has not yet been clinically proven. Centotheca lappacea (L) desv., also known as “Ya Hee Yoom” or “Ya Repair,” belongs to the herbal grass family Poaceae (Gramineae) (3). It has long been used in Thailand and other Asian countries to promote postpartum recovery because it is believed to heal wounds and tighten muscles. For example, postpartum women would smoke the treated area by sitting or standing over a wood-fueled fire and burning C lappacea. Asian cultures have also boiled the grass and consumed the resulting juice to promote rejuvenation and anti-aging. The smoke and residue of the dried grass contains compounds such as phenol derivatives and fatty acids (3). Hot water and ethanol extractions of C lappacea produce extracts rich in phenolic and flavonoid compounds, which have antioxidant and anti-inflammatory properties (3,4). C lappacea extracts exhibit mild tyrosinase and collagenase inhibition, similar to the effects of ascorbic acid. Ethanolic C lappacea extracts exhibit higher antioxidant activities than aqueous extracts, with 1.56-fold more 2,2-Diphenyl-1-Picrylhydrazyl (DPPH) scavenging activity and 1.07-fold more lipid peroxidation inhibition activity. The extract also exhibits no cytotoxicity to human skin fibroblasts at concentrations of 0.1–1,000 μg/mL cell viability remains above 90%. Pro MMP-2 inhibition was 21.49 ± 2.60% and active MMP-2 inhibition was 100 ± 0.00% in fibroblasts treated with C lappacea extract, similar to the inhibition efficacy of ascorbic acid. In addition, the extract demonstrated estrogenic activity at 1,000 μg/mL in MCF-7 cells by enhancing cell proliferation by 107.05 ± 17.36% relative to untreated controls (5). Another study found that C lappacea extracts protect against radiation-induced damage to endothelial cells (6). These findings collectively confirm the traditional use of this plant in postpartum women and support its cosmeceutical development as an anti-aging product. Recent studies have evaluated the bioactivity of C lappacea extracts and formulated the extract into a cream for topical use. Maximum antioxidant activity was achieved with 70% C lappacea extract, which resulted in an IC 50 of 56.02 mg/mL and 199.62 mg/mL as determined by DPPH and 2,2’-azino-bis 3-ethylbenzothiazoline-6-sulfonic acid assays, respectively. Furthermore, this concentration showed the highest total phenolic and flavonoid content (0.42 mg gallic acid/g extract and 0.24 mg catechin/g extract, respectively). As a result, 70% C lappacea extract was evaluated for anti-tyrosinase and anti-collagenase activities. 70% C lappacea extract inhibited collagenase activity with an IC 50 of 30.78 mg/ mL, but the IC 50 for anti-tyrosinase activity could not be determined. The stability of 70% C lappacea extract was determined by comparing initial antioxidant activity, as measured with the DPPH assay, to antioxidant activity after 3 months of storage at 4, 25, and 40°C. Antioxidant activity was not significantly reduced, regardless of the storage temperature. Cream containing 70% C lappacea extract at a final concentration of 0.5% was stable at all storage conditions (7). Despite their potential promise, C lappacea extract-based creams have not yet been tested in clinical trials. This study therefore aimed to evaluate the efficacy of a new facial cream
5 COMPARATIVE CLINICAL EVALUATION OF C LAPPACEA formulated using C lappacea. The effects of the new cream on skin lightening, firmness, and elasticity were evaluated using noninvasive measurements. MATERIALS AND METHODS RESEARCH DESIGN This randomized, double-blind, 5 week, pre- and post-comparison clinical trial was conducted from September – October 2017. This study was conducted in compliance with the ethical principles of the Institutional Review Board (IRB) and Institutional Ethics Committee of the Institute of Dermatology, Bangkok, Thailand. The study design was reviewed by the appropriate ethics committees and approved for clinical implementation (Study Code IRB/IEB 002/2561). All research was conducted according to the ethical principles of the Helsinki Declaration (1964). This study included 44 Asian females ages 30–55 with at least 1 facial melasma site. The subjects were fully informed about the purpose of the study and provided written consent. Exclusion criteria included (1) pregnant or breastfeeding, (2) taking oral contraceptives or hormone treatment, (3) a history of cancer or skin cancer, (4) use of topical anti-aging treatment (including anti-aging products, whitening substances, bleaching agents, topical retinoids, and alpha- or beta-hydroxy acid) within 2 months preceding the screening visit, and (5) treatment with aesthetic lasers, Botox, filler, or thread lift within 6 months preceding the screening visit. Subjects with other facial conditions that could potentially affect the study results were also excluded. Subjects with a history of allergic reactions to cosmetic products, sunscreens, or ingredients in the tested product were not recruited for the study. TEST PRODUCT Subjects were randomly assigned to apply either C lappacea cream or a control (placebo) cream to their entire face twice daily for 4 weeks. The test (labeled A) and placebo (labeled B) creams were packaged in identical 2 g tubes, for twice daily use. The test product was composed of C lappacea extract (0.1%) with other ingredients (Appendix) and broad- spectrum ultraviolet A and ultraviolet B filters ethylhexyl methoxycinnamate, octocrylene, butyl methoxydibenzoylmethane, and titanium dioxide were included to provide sun protection at SPF 30. The control product was identical but excluded the C lappacea extract. PRODUCT APPLICATION Subjects were instructed to apply the test or control cream to the entire face twice daily (morning and bedtime) for 4 weeks. After washing the face with gentle liquid soap (provided), subjects gently squeezed ½ tube of cream (approximately 1 g) onto the back of their hand, which they then distributed across their face in five dots (forehead, nose, two cheeks, and chin) using a fingertip. On the forehead, nose, and chin, subjects gently massaged upward until the product penetrated the skin. On cheeks, subjects gently massaged in a circular direction, from the lower cheek to the upper cheek, until the product penetrated the skin. Sunscreen (provided) was thoroughly applied on top of the cream. All subjects were asked
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)