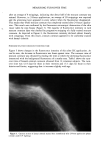
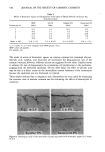
POLYMERS AND LIPIDS ON SKIN BY ESCA 299 baseline against which to compare the surface composition produced by exposure to various conditioning agents. Deposition of these polymers can be established by several parameters, the first of which is the C/O ratio. All three polymers tested contain a much higher percentage of oxygen than does the native skin surface. Therefore, polymer adsorption causes a decrease in the C/O ratio relative to that observed on the untreated controls. This increased oxygen content is also observed in the carbon high-resolution spectra since the oxygen in these polymers is predominantly in the form of alcohol and ether linkages. As shown in Figure 3, adsorbed polymer is thus reflected as increases in the 286.1 eV carbon peak characteristic of alcohol/ether-type carbon. The final indi- cator of polymer deposition is also found in the high-resolution data. Each of the polymers tested contains cationic quaternary nitrogen groups. This quaternary nitrogen can be distinguished from the native amide form (Figure 4), thus serving as a tag for adsorption. Tables III and IV summarize the data obtained from polymer-treated membranes which were not washed in SDS prior to polymer treatment. These samples displayed inter- esting behavior in that adsorption of the polymers was detected on the inner membrane surface (as evidenced by the presence of the quaternary nitrogen as shown in Table IV) however, only Polyquaternium-24 appeared to be adsorbed to any significant degree on the outer skin surface. The adsorption behavior on the outer skin surface of these samples is interpreted as a manifestation of the greater hydrophobicity, and hence lipo- philicity, of the Polyquaternium-24 molecule (5). As evidenced by the data on the control skin surfaces, the unwashed skin surface has a substantial lipid content. The Table II High-Resolution Results of Control Skin Samples Atomic % C N Treatment 284.6 286.1 287.6 288.9 399.6 402 Untreated controls Outer side Inner side SDS-washed controls Outer side Inner side X 65.4 10.0 3.5 2.2 2.3 N.D. S 3.4 1.3 0.9 0.5 0.9 -- X 64.3 9.1 4.0 2.4 3.0 N.D. S 0.6 0.9 0.6 0.3 1.0 X 52.5 12.8 6.4 2.1 8.3 N.D. S 0.6 0.5 0.7 0.4 0.3 -- X 50.3 12.6 6.9 2.7 9.0 N.D. S 1.8 0.6 1.0 0.9 0.2 N.D. = not detected. Assignments: C 284.6 C- C hydrocarbon 286.1 C- O alcohol/ether 287.6 N-C=O amide 288.90 - C = O acid/ester N 399.6 amide 402 NR4 +
300 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 299.8 C-C c.o } 279.8 BINDING ENERGY {eV) Figure 3. Carbon high-resolution spectrum of A) unwashed control skin, and B) skin treated with Poly- quaternium-24. chitosan and Polyquaternium-10 molecules either do not appear to be attracted to this lipid-covered surface or are readily removed in the subsequent distilled water rinse. The data on the inner membrane surface appears to be contradictory in light of this proposed mechanism for deposition. However, the previously cited ESCA study of the action of various organic solvents and surfactants showed that skin lipid removal was much more facile from the inner skin surface than from the outer skin (3). The pro- AMIDE NR4+ // • III III % ß i II •.o BINDING ENERGY (eV) 3•.0 •/•ure 4. Nitrogen hi•h-reso]ution spectrum oF Polyqu•ternium-10-tre•ted skin s•mple.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)