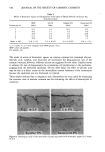
PROVITAMIN TO C AND E IN SKIN 337 360 0 12 24 36 48 60 72 Time Figure 3. Cumulative amount o• v•i• C Noco•ver•ed {ro• until the concentration in the stratum corneum reaches a steady state. This process may require a substantial period of time. The cumulative amount of the provitamin EPC-Na appearing in the receptor solution is shown in Figure 5. It was found that the concentration of provitamin increased linearly after the lag time of about 24 hours. This finding indicates that the provitamin was not completely bioconverted to vitamins C and E in the hairless mouse skin. The steady- state rate of appearance of intact provitamin was calculated to be 1.4 x 10 -3 p•mole/ cm2/hr from the slope of the linear portion of the permeation profile. Based on the 4O •.. 30 lO 0 24 48 72 Time (hi Figure 4. Cumulative amount of vitamin E bioconverted from EPC-Na and appearing in the receptor solution. The circles and error bars represent the average and standard error of six data points.
338 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 50- 40 • 30 c• 20 10 I O0 - 24 48 72 Time (h) Figure 5. Cumulative amount of EPC-Na permeated across the skin. The circles and error bars represent the average and standard error of six data points. steady-state rate of permeation of EPC-Na and the steady-state rate of appearance of vitamin C, the yield of bioconversion was calculated to be 96% in the hairless mouse skin. It is obvious, however, that the yield of the provitamin bioconversion is in- fluenced by the activity and distribution of enzymes in the skin therefore, the findings obtained from the hairless mouse may not be directly applicable to man. Suffice it to say, however, that the provitamin, EPC-Na, will be bioconvertible to vitamins C and E simultaneously in the human skin because of the enzyme (esterase) distribution in human skin (17). ACKNOWLEDGEMENTS We express our sincere thanks to Senju Pharmaceutical Co. Ltd., Osaka, Japan, for providing EPC-Na. We are also grateful to Drs. Y. W. Chien and O. Siddiqui for their valuable comments. REFERENCES (1) L. J. Machlin, in Handbook of Vitamins (Marcel Dekker, New York, 1984). (2) C. J. Bates, in Vitamin C (Astorbit Acid), J. N. Counsell and D. H. Hornig, Eds. (Applied Science Publishers, Englewood, NJ, 1982), pp. 1-22. (3) D. Djerassi, Vitamin E: Biochemical function and its role in cosmetics, D&CI, 46-77 (March 1986). (4) K. Furuse, Vitamin E--Biochemistry and biochemical function, Pharm. Tech. Japan, 2, 729-735 (1986). (5) R. E. Keith, B. M. Chrisley, and J. A. Driskell, Dietary vitamin C supplementation and plasma vitamin E levels in humans, Am. J. Clin. Nutr., 33, 2394-2395 (1980). (6) E. Ginter, A. Kosinova, A. Hudecova, and A. Madaric, Synergism between vitamins C and E: Effect on microsomal hydroxylation in guinea pig liver, Internat. J. Vit. Nutr. Res., 52, 55-59 (1982). (7) L. J. Machlin, in Vitamin E (Marcel Dekker, New York, 1980).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)