-• J. Soc. Cosmet. Chem., 38, 333-339 (September/October 1987) Bioconversion of a provitamin to vitamins C and E in skin KAKUJI TOJO and AE-RI C. LEE, Department of Pharmaceutics, William Levine Hall of College of Pharmacy, Rutgers--The State University of New Jersey, Busch Campus, Piscataway, NJ 08855-0789. Received May 4, 1987. Synopsis Skin bioconversion of a new provitamin, dl-o•-tocopherol-L-ascorbic acid sodium phosphate diester, for vitamins C and E, was investigated using a hairless mouse skin in vitro. The provitamin, which is quite stable in aqueous solutions, was efficiently bioconverted to both vitamins in the viable skin of hairless mice. The appearance rate of vitamin C, after the provitamin bioconversion, reached a steady state very quickly and virtually no time lag was observed. The appearance rate of vitamin E, after the bioconversion, increased gradually during the entire period of the permeation and bioconversion experiment (72 hours), after a long time lag (about 24 hours). The long time lag indicates that vitamin E may bind strongly to the skin tissue. From the steady-state appearance rates of vitmin C and the provitamin, the yield of the skin bioconversion was found to be 96% in the hairless mouse skin. INTRODUCTION Ascorbic acid (vitamin C) and o•-tocopherol (vitamin E) are two essential vitamins. An adequate supply of these vitamins is needed to preserve healthy skin as well as to avoid various deficiency diseases (1). Vitamin C plays an important role in several oxidation reactions. It is believed that this vitamin is essential for hydroxylation of prolyl and lysyl residues in growing chains of hydroxylareal procollagen peptide in the skin (2). Vitamin E, on the other hand, has been applied topically for many years as a natural moisturizer. In recent studies, various therapeutic efficacies of vitamin E have been revealed (3,4). Vitamins C and E were found to function in a synergistic fashion to protect against the peroxidation of membrane lipids (5,6). Vitamin E is a lipophilic compound (octanol/water partition coefficient = 480), while vitamin C is a hydrophilic compound (partition coefficient = 0.02) which is hardly soluble in lipophilic solu- tions. Vitamins C and E are not stable in many solutions (1,7), although the stability may be enhanced by using various stabilizing agents (8). Since the skin is a highly heterogeneous membrane which consists of lipophilic stratum comeurn and lipophobic viable skin, the skin permeation of a drug depends on the hydrophilicity or lipophilicity of the penerrant. In recent years, the percutaneous absorption of various drugs has been studied exten- sively to develop either topical or systemic drug delivery (9- 11). Topical application of 333
334 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS vitamins has also been investigated by many researchers (12- 14). However, neither the dynamics nor the steady state permeation of these vitamins across the skin has been studied quantitatively. In this communication, the skin permeation and bioconversion of a new provitamin for vitamins C and E is described using an in vitro skin permeation apparatus (15). The hairless mouse skin (HRS/J strain (16)) is used in this study not only because this animal skin is well controlled in terms of sex, age, weight, and thickness, but also because the skin contains active enzymes (esterase) for the biocon- version of prodrug esters (16) which are also found in the hum'an skin (17). MATERIALS AND METHODS The provitamin for vitamins C and E, dl-c•-tocopherol-Lmscorbic acid sodium phos- phate diester (EPC-Na, Figure 1), was synthesized and supplied by Senju Pharmaceu- tical Co. (Osaka, Japan). EPC-Na is easily dissolved and is stable in aqueous solutions such as water or saline. The solubility of EPC-Na in water was found to be 384 mg/ml. It is expected that EPC-Na is bioconverted to vitamins C and E in the presence of active enzymes (esterase) in the skin, as illustrated in Figure 2. All chemicals were of reagent grade. All solutions were prepared with deionized distilled water which had been de- gassed before use. The provitamin (EPC-Na) and vitamins C and E were assayed by HPLC. Since we were not able to assay each compound simultaneously under a single HPLC separation condi- tion, we repeated the same skin permeation experiment three times to measure the rates of appearance of the compounds in receptor solution. In each permeation experiment, we used six sets of the skin permeation systems (15). All experiments were carried out ?=0 CHa C-OH H•C • -O CH• ?Ha ?•-OH •(CH2CH•,CH•, C H)a-CHa •iCi H HO I CHa CH-OH I Vt. C Vt. E Figure 1. Provitamin for ascorbic acid (vitamin C) and a-tocopherol (vitamin E), dl-a-tocopherol-L- ascorbic acid sodium phosphate diester (EPC-Na).
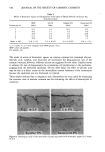
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)