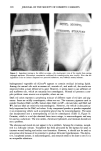
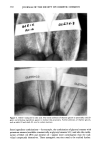
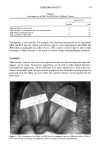
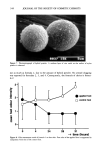
COMEDOGENICITY 331 Table II Comedogenicity of D&C Red #36 Dye in Different Vehicles Grade (0- 5) Comedo. lrrit. D&C red #36 in mineral oil D&C red #35 in pentaerythrital tetra caprdcaprylate D&C #36 in propylene glycol D&C red #36 in PEG 400 3 0 2 0 1 0 0 0 The opposite is also possible. For example, the combination produced by the ingredient D&C red #36 and the vehicle polyethylene glycol is less comedogenic than D&C red #36 when incorporated into other vehicles. The cosmetic chemist may be able to take advantage of these findings in the future to custom design noncomedogenic products. SUMMARY These studies indicate that skin care preparations that are nonirritating and noncome- dogenic can be made. Nonreactive ingredients can be used to make elegant products, and borderline ingredients can be combined with other ingredients to reduce the reac- tions to acceptable levels. In spite of these guidelines, new formulations must always be examined with the rabbit ear assay before the cosmetic chemist can be assured that his ideas work. Figure 5. The comedogenicity of D&C red #36 when incorporated into two different vehicles. The ve- hicle may increase or decrease an ingredient's ability to produce follicular hyperkeratosis.
332 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table III Effects of the Solvent on Comedogenicity and/or Irritancy of Fatty Acids Organic solvent* Sunflower oil Grade (0- 5) Grade (0- 5) Fatty acids Comedo. Irrit. Comedo. Irrit. Caproic acid 0 4 2 2 Caprylic acid 1 3 1 1 Capric acid 2 2 3 1 Lauric acid 3 1 4 1 Myristic acid 1 0 3 0 Palrnitic acid 0 1 2 0 Stearic acid 0 1 2 0 Archidic acid 1 1 2 0 Behenic acid 1 0 1 0 * Ethyl ether or acetone. The rabbit ear assay remains important to the rapid evaluation of new ingredients and the cosmetic chemist's formulations. Both the visual and microscopic evaluations of the rabbit ear need to be done simultaneously (9). Materials found to be noncomedogenic in the rabbit assay appear to be noncomedogenic in the human model (10). Whether highly comedogenic ingredients in the rabbit ear assay are always comedogenic in humans still remains uncertain. Currently, it is more prudent to avoid these offenders. The major offenders, such as isopropyl myristate, acetylated lanolin alcohol, and lauric acid derivatives such as laureth-4, should be used with caution in skin care products. We are not convinced of the statement that lower concentrations of these compounds can be safely used with no comedogenic consequences (11). Human skin studies have been used to give that statement credence, but the back skin of human volunteers is relatively insensitive (7). However, when the rabbit ear assay is positive but the human back skin results are negative after only eight weeks' exposure, the results from the rabbit ear assay should not be dismissed. The reaction may take longer or the back skin may not be the ideal testing surface. An additional "bonus" of the rabbit ear assay is detection of the potential of an ingre- dient or finished product to produce an epithelial irritant reaction. It is easy to keep track of the surface irritancy while doing the follicular studies. The stratum corneum of the rabbit ear is very thin and undeveloped. This results in an extreme sensitivity of the skin to exposure to irritants. If this test finding is confirmed by others, we may find it unnecessary to use the Draize rabbit dermal irritancy test. This paper is meant to be a survey of the ingredients used in skin care and hair care products. The survey is not at all definitive but simply designed to stimulate research, so that new noncomedogenic products will become available for those of us with acne- prone complexions. This subject has recently received an excellent review by the Amer- ican Academy of Dermatology Invitational Symposium on Comedogenicity (12). REFERENCES (1) A. M. Kligman and O. H. Mills: Acne cosmetica, Arch. Dermatol., 106, 843-850 (1972). (2) J. E. Fulton, S. Bradley, et al, Noncomedogenic cosmetics, Cutis, 17, 344-351 (1976).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)