360 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS We speculate that PHB may be a classical chelating agent, in the sense described by Marshall et al. (29). Intracellular accumulation of PHB by P. cepacia (24) may enable this species to retain divalent metal ions as a PHB chelate, which could provide an internal reservoir that may help prevent loss of metal ions to exogenous chelators. This would enable P. cepacia to resist more effectively the destabilization caused by external chelating agents (7) than do the fluorescent pseudomonads, which do not accumulate PHB (24). Definitive studies are needed to confirm this. The data in Table II show that the preservative system in this lotion was less effective for S. aureus, B. cereus, and E. coli than it was for many of the Pseudomonas test cultures. The percentage of sporulation of Bacillus sp. was 30-50% at 24 hr (8) however, B. cereus produced only a few visible spores in a microscopic field (1000 X ) when 24-hr TSALT cultures were suspended, stained, and examined microscopically. Experience with Bacillus sp. and B. cereus 11778 revealed that these organisms produce few (if any) preservative system-resistant spores during 24-hr growth on TSALT at 37øC. These organisms were used to determine the effects of preservative systems on vegetative ba- cilli. The data in Table III illustrate anti-Pseudomonas synergy because the rate of inactivation (i.e., slope of the survivor curve) of each population of test organisms in MP + 934, 941, or 1342 was a larger negative number than the sum of the rates (slopes) of inacti- vation in MP and each acrylic acid homopolymer/copolymer taken separately. P. cepacia showed an additive effect in lotion containing MP + 1342. The effect of nonionic lotion pH on the results of preservative efficacy testing with P. aeruginosa, P. cepacia, P. fluorescens, and P. putid• was determined in lotions adjusted to pH 6-9 by adding varying amounts of TEA. No consistent effect of lotion pH on antibacterial activity with these four test organisms was observed. The antibacterial effect of MP is reported to increase with decreasing pH below the pKa of MP (pH 8.17) (30). P. cepacia was inactivated more slowly than the other pseudomonads in lotions adjusted to pH 6-9. Incorporation of •0.1% 934, 941, or 1342 into the nonionic lotion produced a marked decrease in the D-values for P. aeruginosa, P. cepacia, P. fluorescens, and P. putida, com- pared to the D-values obtained in lotions containing no 934, 941, or 1342. We were unable to demonstrate a consistent relationship between the acrylic acid homopolymer/ copolymer concentration, from 0.1-0.4%, and the observed rates of death of the test organisms. It is possible that the maximum synergistic action was obtained at •0.1% polyacrylic acid/acrylic acid copolymer so that higher concentrations produced no fur- ther increase in anti-Pseudomonas activity. The addition of 0.1% CaCi 2 to the nonionic lotion containing 0.2% 1342 and 0.2% MP produced significant increases in the D-values for the fluorescent pseudomonads (P. aeruginosa, P. fluorescens, and P. putida) and eliminated the anti-Pseudomonas synergy. The opposite effect was observed with P. cepacia, because addition of 0.1% CaCi 2 pro- duced a significant decrease in D-values for this organism (Table IV). The inhibitory effects of CaCI 2 on P. cepacia may have been due primarily to the decrease in the pH of this lotion caused by the addition of CaC12, compared to the control. P. cepacia was the only test organism that did not show synergistic anti-Pseudomonas activity in the pres- ence of 1342 and MP (Table III). These results reflect the physiological diversity of different species of Pseudomonas.
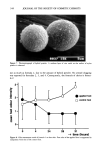
SYNERGY OF PRESERVATIVES 361 Analyses of tap water and 1342-treated tap water indicated that exposure of the water to the 1342 caused a significant decrease in hardness, expressed as ppm CaCO 3. This suggests that 1342 is capable of chelating Ca 2+ ions. The 0.1% 1342 and 0.01% Na2EDTA reduced water hardness by similar amounts. This suggests that these com- pounds have similar chelating abilities for the Ca 2 + ions. The ST study revealed little antibacterial activity by either MP or Na2EDTA alone (Table V). Rapid killing occurred in the presence of MP + Na2EDTA because no viable organisms were recovered at 4 hr in most test systems. The test organisms found to be more persistent in these tests were, in general, more persistent in lotions con- taining 1342 and MP (Table II). In some cases, it is believed that differences in results between these two tables may be attributed to differences in APCs of the inocula. The STs and MPSTs in Table V were used to calculate the slopes and D-values (or MPSlopes and MPD-values) in Table VI. Survivor curve slopes may be determined when the STs and initial inocula of the test organisms are known. For example, ?. aeruginosa 9027 had an ST of 4 hr in MP + Na2EDTA, and the APC in the sample was 1.1 X 105/ml. Here, the D-value and slope were 0.8 hr and - 1.26 hr- •, respectively. Where STs are not known (i.e., ST 48 hr), the MPD-values and corresponding MPSlopes may be estimated from a virtual survivor curve constructed using the APC of the inoculum and the MPST, as explained above. Here, the MPD-value for P. aeruginosa 9027 in MP was calculated to be 9.5 hr and the MPSlope was - 0.105 hr- 1. This slope is the negative reciprocal of the MPD-value and represents the fastest possible rate of death of this organism in this test system. If P. aeruginosa were being killed at a faster rate, then no organisms would have been recovered at the last sampling (i.e., at 48 hr). The MPSlope for P. aeruginosa 9027 in Na 2 EDTA was estimated similarly to be - 0. 105 hr-1 Synergy was observed here, because the slope for the system con- taining MP + Na2EDTA (- 1.26 hr-1) was a larger negative number than the sum of the MPSlopes for MP and for Na2EDTA (- 0.210 hr-1). This procedure was used for each test organism shown in Table VI. The MP q- Na2EDTA system had synergistic anti-Pseudomonas activity for all pseudomonads, except for P. cepacia 13945. The MPD- values for different strains of P. aeruginosa were slightly different due to the slightly different concentrations of organisms in the inocula (Table VI). The estimated STs for S. aureus and E. coli were 48 hr in all test systems consequently, it was not possible to establish synergy for these organisms in this experiment. Numerous workers have reported the enhancement of preservative action by EDTA (3,7,31-34). The potentiation by EDTA is believed to be due to permeabilization synergy, in which one antimicrobial agent (EDTA) assists the passage of the other antimicrobial through the cell wall or membrane (7). We propose that the anti-Pseudo- toohas synergy observed with 934, 941, or 1342 and MP is due, at least in part, to chelation of divalent metal ions and that it is similar to permeabilization synergy re- ported for the potentiation of preservative action by EDTA (7). Results in support of this are the demonstration that 1342 has chelation activity, the elimination of the synergism observed with the fluorescent pseudomonads by the addition of 0.1% CaC12 (Table IV), and the similarities in the survival patterns of the various pseudomonads in nonionic lotions with polyacrylic acid or acrylic acid copolymer/MP systems (Table II) and in Na2EDTMMP solutions (Table V). Similar patterns of inactivation were observed in both aqueous and nonionic emulsion
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)