j. Soc. Cosmet. Chem., 41, 347-358 (November/December 1990) Study of lipid and non-lipid structure in human stratum corneum by X-ray diffraction J. c. GARSON, J. DOUCET, G. TSOUCARIS, and J. L. L•V•QUE, Laboratoires de Recherche de L'Oreal, Aulnay sous Bois (J.C.G., J.L.L.), LURE, Bat. 209 C, Universitg de Paris Sud, Orsay (J, D. ), and CNRS, UPR, Universitg de Paris Sud, Chatenay Malabry ( G. T, ), France Received September 21, 1990. Presented at the IFSCC Meeting, New York, October 1990. Synopsis The molecular organization of human stratum corneum (SC) is still poorly known, in spite of its importance for the understanding of its biological functions. By using ø synchrotron X-ray diffraction and oriented samples, new basic features were revealed in the 2.5-to-125 A domain. The X-ray patterns were richer and more complex than previously published. Lipid extraction allows discrimination between the proteic or lipidic origin of the numerous arcs and rings observed. Concerning the proteins, we found that the keratin organization (or versus {3) in human stratum corneum depends on its origin and environment, contrary to the commonly accepted ot form for skin keratin. For common SC it would be close to {3, and for "callus" type it is or. An unknown {3-pleated sheet protein with an 9.4 • interchain distance was inferred from the diffraction data. Reticular distances of the lipidic lameliar organization were evidenced by small-angle diffraction (10-65 •). Wide angle diffraction (2.5-10 •) showed intermolecular distances of different species of lipids. Certain features of the X-ray diagrams can be assigned (i.e., cholesterol). Liposomes, which are made also of organized lipids, can be characterized by an X-ray diagram. This fact is important for cosmetic research, providing the possibility to study the interaction between the stratum corneum and liposomes. Some examples will be reported. INTRODUCTION X-ray diffraction is the method of choice for the study of the spatial organization of macromolecules in biological tissues. This technique has been used for a large number of years in the investigation of atomic structure in metals and alloys and is a basic research tool in crystallography. In the field of biology, interest in X-ray diffraction increased when Watson and Crick discovered the double-helix form of DNA for which they were awarded the Nobel prize. Prior to this work, the diffraction patterns of keratins obtained in 1933 by Astbury marked the beginnings of molecular biology. 347
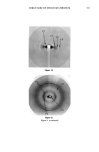
348 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS With regard to the skin, the quality of the patterns obtained in the stratum corneum (SC), for example, has been rather poor, mainly due to the fact that this keratinized membrane is of a composite nature, possessing both amorphous and crystalline zones that confer a poorly organized pseudosolid character. In addition, in order to obtain reasonably clear diffraction patterns in the SC, high doses of X-rays are used, requiring very long times of exposure if normal sources are used. However, certain studies of SC specimens of different types have been conducted re- cently and concern the organization of keratin (1-3) and lipids (4-6). So far, it has not been possible to determine whether keratin is organized in the ot or [3 form, due to the diversity of the SC specimens and the poor quality of the diffraction patterns obtained. With regard to lipid organization, apart from the results of White obtained in hairless mice, the patterns are very disappointing. They do, however, show an organization in bilayers, confirming the arrangements of polar lipids forwarded by Elias (7) and recently demonstrated by Swartzendruber (8). This form of lipid organization is of great interest, since according to numerous au- thors, it forms the basis of the barrier function of the stratum corneum (9). Lipids would also appear to be involved in the mutual adhesion of corneocytes and in the plasticity of the horny layer (10,11). We therefore studied the organization of proteins and lipids in the stratum corneum by means of X-ray diffraction, paying particular attention to the exact form (or or [3) of keratin in various SC specimens and the supramolecular organization of intercellular lipids. The observed intercellular lipid organization was compared with that found in liposome vesicles commonly used in cosmetics. In order to overcome the difficulties mentioned above (relative to the quality of the X-ray diffraction patterns obtained with the human SC), we used a high-energy X-ray source (synchrotron). MATERIALS AND METHODS EXPERIMENTAL SET-UP The X-ray beam was generated by the LURE synchrotron at the Universit• de Paris Sud. A simplified diagram of the equipment is given in Figure 1. In comparison with a conventional source, the intensity and resolution obtained can be increased by a factor of 100 and 5, respectively. A wavelength of 1.61 • was chosen. The specimens were placed on a goniometric head mounted on a multi-axis holder. A coincidental optical system enables the specimen to be oriented precisely relative to the beam axis. The distance between the specimen and the film can be varied in such a way as to obtain reticular planes from 2 to 70 •. Using another chamber, reticular planes from 20 to 200 • can be explored. The specimens placed in the chambers consist of a number of sheets of SC (2 to 16) that can be oriented either perpendicular or parallel to the beam axis. The SC sections (5 mm X 15 mm) are kept in place by means of specimen holders.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)