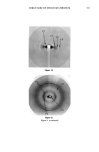
STRUCTURE OF STRATUM CORNEUM 349 Other experiment Linear accelerator •- e + Monochromator •nchrotron radiation Film SCHEMATIC OF THE EXPERIMENTAL SET-UP Figure 1. Schematic of the experimental set-up. SC SAMPLING The human SC specimens used were obtained during plastic surgery. The epidermis was separated from the dermis by treatment with humidified air at 56øC. The SC was separated from the epidermis by trypsin treatment. LIPOSOMES The vesicles studied were obtained by agitating a mixture of nonionic lipids, choles- terol, and dicethyl phosphate (47.5/47.5/5). Transmission electron micrographs of the vesicles were obtained after indirect staining. RESULTS AND DISCUSSION PROTEIN COMPONENTS The results concerning the supramolecular organization of the protein components in the SC are given in Table I and Figure 2. This preliminary identification was obtained by the delipidization of SC using a chloroform/methanol mixture and comparison with untreated specimens. As seen in Table I, three groups of reticular distances can be distinguished, corresponding to three different aspects (diffuse zone, sharp arc, and broad band). It is difficult to interpret the first group, P 1, since it is situated in a diffuse zone. The values obtained for this group nonetheless confirm those reported by White for SKH mouse SC. Group P2 corresponds to the fundamental and harmonics of a fine arc situated at 9.4 •. This sharp arc represents the organizatioon of a protein on a domain sufficiently large to attribute a size of approximately 3000 A. (Scherrer's formula is used to determine the
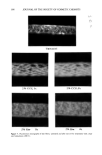
350 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table I Stratum Corneum Proteic Scattering Components Group d (•) Aspect Intensity P ! 125 // Equatorial diffuse zone vs 125 // Equatorial diffuse zone vs 86 // Equatorial diffuse zone vs P2 9.4 // Equatorial sharp arc vs 4.7 // Equatorial sharp arc s 3.15 // Equatorial sharp arc w P3 9.5 //3, Nonisotropic broad band vs 4.58//l Isotropic broad band s Intensity: vs, very strong s, strong w, weak. size of a diffracting object from the sharpness of the arc). This protein would appear to have a planar structure of type 13, as the value of 9.4 • is typical of the inter-chain distance of type-13 forms (12). It is also difficult to determine the origin and nature of this protein constituent, which has previously been reported by White in the SKH mouse SC. The size of the dif- fracting domain is, however, larger than that of the intercellular spaces this would suggest that the protein is an intracorneocyte protein such as filaggrin, which has a partial 13 structure. However, the desmosome remnants are situated in dilated zones of the SC, and such an origin cannot be excluded for this unidentified molecule. The confirmation of this hypothesis would be very important given the cohesive role of these elements between corneocytes. Figure 2a Figure 2. X-ray patterns of normal stratum comeum sample: a. Pattern in geometry 3_ b. Pattern in geometry// c. Pattern in geometry//.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)