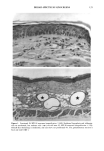
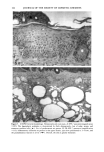
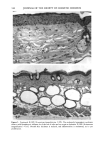
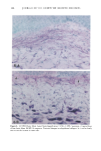
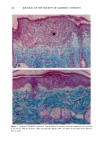
118 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS radiation (4). Alcalay et al. reported that low-dose UVB radiation (between 0.75 and 1.5 MED) decreased the number of ATPase-positive epidermal Langerhans cells to between 21% and 65% (5). In animal studies, UVB-induced reduction of Langerhans cell density was observed in a dose-dependent manner (6,7). However, UVB radiation did not decrease the number, but impaired the function of Langerhans cells in vitro (8,9). Much less is known about the effect of chronic sun exposure. Thiers et al. studied an older population with clinical evidence of sun-damaged skin and compared the skin of the arm and the buttock regions (10). They concluded that chronic sun exposure resulted in a decrease of ATPase-positive Langerhans cells. Gilchrest et al. reported that the number of Langerhans cells in sun-exposed areas was statistically significantly fewer than in paired sun-protected specimens (11). Delo et al. reported that the density of Langerhans cells in chronic actinically damaged skin at the posterior neck was significantly decreased compared to that of control skin (12). In this study, we investigated the effect of chronic actinic exposure on Langerhans cell density not in a photodamaged skin but in a normal skin, since the Langerhans cells in the photodamaged skin might be decreased by the secondary effect of the other derreal damage. Thus, the relatively limited data for normal skin and the availability of mono- clonal antibodies for more specific immunohistochemical identification of Langerhans cells prompted us to carry out this study ofdendritic cell populations in sun-exposed and sun-protected skin from young and elderly individuals of two ethnic groups. Another reason for the present study was the limited baseline data on Langerhans cells in normal human skin from individuals of different age ranges and ethnic backgrounds. Studies designed to assess age-related changes include that of Gilchrest et al. (4), who showed an age-associated decrease in the population of Langerhans cells in Caucasians by comparing four young and seven old subjects, both male and female. Thiers et al. (10) showed a significant decrease in the number of ATPase-positive Langerhans cells in skin from elderly patients when compared to that from young medical students. Gilhar et al. reported that the number of HLA-DR positive Langerhans cells in sun-protected thigh skin decreased with age (13). As for animals, a reduction of Langerhans cells in aged murine ear epidermis was reported (14), and the Langerhans cell density of C57BL/6J mice was found to decrease with age (15). With regard to ethnic background, Berman et al. found no differences between the mean densities of Langerhans cells in skin from male or female Caucasians and Hispanics (16). However, no baseline data for the Langerhans cell density in African-Americans is available at present. From the previous epidemiological study, UV-induced skin cancers are rare among African-Americans. Therefore, it is intriguing to investigate the effect of chronic sun exposure on the Langerhans cell density in African-Americans. MATERIALS AND METHODS STUDY SUBJECTS Thirty-seven female volunteers (13 Caucasians aged under 30 years, ten Caucasians aged over 60 years, seven African-Americans aged under 33 years, and seven African-Amer- icans aged over 59 years) participated in the main study. One Caucasian male volunteer with a clinically photodamaged skin also participated in the study. All subjects enrolled
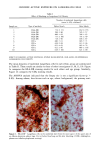
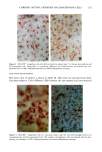
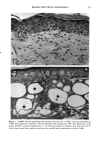
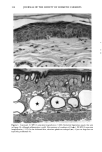
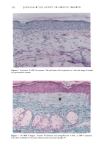
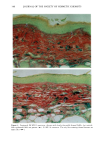
CHRONIC ACTINIC EXPOSURE ON LANGERHANS CELLS 119 were in good health with no record of abnormal reactions to sunlight. Before the project started, informed consent was obtained after the nature of the procedures had been fully explained. The subjects did not take any medication during the study. BIOPSY PROCEDURE A shave biopsy was taken from the inner aspect of the upper arm and the outer aspect of the forearm after local anesthesia with lidocaine. The inner aspect of the upper arm was considered to be a site without chronic sun exposure. In the main experiment, the sun-exposed site (outer aspect of forearm), biopsied during the winter and early spring, had not been exposed to sunlight within one month prior to biopsy in order to eliminate any acute effect of sunlight exposure. However, the sun exposure had not been con- trolled for the subject with photodamaged skin, and the biopsies were taken in the summer. EPIDERMAL SHEET PREPARATION The shave biopsy was immersed in 0.5 M NH4SCN tris-buffered saline for 20 minutes at 37øC. The epidermal sheet was separated from the dermis with fine forceps and rinsed with tris-buffered saline at pH 7.6. The epidermal sheet was then fixed in acetone for ten minutes and rehydrated in tris-buffered saline after fixation. The fixed tissue was immediately stained or held for up to three days in tris-buffered saline at 4øC prior to immunohistochemical staining. IMMUNOHISTOCHEMICAL STAINING Since HLA-DR and CDla markers are specific to Langerhans cells in human epidermis (17), we used mouse monoclonal anti-HLA-DR and anti-CDla antibodies as the primary antibodies, with a standard alkalinephosphatase technique as the staining method. Anti-HLA-DR antibody (DAKO Corporation, Carpinteria, CA) was used as a 1:60 dilution in tris-buffered saline, and anti-CDla antibody (Ortho Diagnostic Systems, Raritan, NJ, or DAKO Corporation) was used as a 1:5 dilution in DMEM with 10% bovine serum albumin. In the staining process the tissue was incubated with anti-HLA-DR antibody for 30 minutes at room temperature or with anti-CDla antibody for one hour at 37øC. The alkalinephosphatase-anti-alkalinephosphatase (APAAP) stain- ing was then performed using the DAKO Universal APAAP KIT System 40 TM. After a brief rinse in tris-buffered saline, the epidermal sheet was incubated with rabbit anti-mouse antibody. The tissue was then treated with the APAAP complex, and the color was developed using Fast Red solution as the chromogen (18). The tissue was mounted dermis side up on standard glass slides using a hydrophilic mounting medium (Glycergel TM, DAKO Corporation). Highly pigmented specimens were bleached by the following method if necessary: after staining, the tissue was immersed in 0.25% KMnO 4 solution at room temperature for ten minutes, rinsed in running water, then immersed in 5 % oxalic acid solution for 40 seconds, rinsed in running water, and placed in tris-buffered saline (pH 7.6) for ten minutes prior to mounting.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)