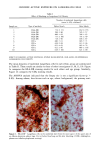
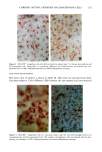
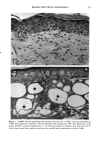
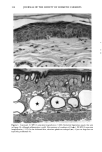
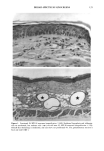
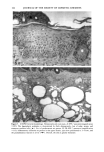
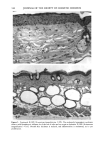
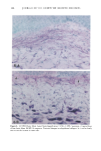
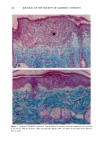
j. Sac. Cosmet. Chem., 47, 167-175 (May/June) A new cell culture method for phototoxicity testing' Effects of oral hypoglycemic and diuretic agents EDGAR SELVAAG, HELLE ANHOLT, JOHAN MOAN, and PER THUNE, Department of Dermatology, Ullevaal Hospital (E.So, P. T. ), and Department of Biophysics, Norwegian Radiumhospital, Montebello (H.A., J.M. ), University of Oslo, Oslo, Norway. Accepted for publication July 18, 1996. Synopsis Cervical cells from human in situ carcinoma, the NHIK (Norsk Hydro's Institute for Kreftforskning) 3025 cell, were utilized for screening phototoxic drugs. Sulphonamide oral antidiabetics and diuretics were incubated with the cells for one hour and then irradiated with broad-band UV and UVA. Cell death was dependent on UVA-fluence and drug concentration. Phototoxicity was demonstrated in the presence of two oral antidiabetics and ten diuretics. A number of substances, not previously described to exert phototoxic effects, were detected in this model. Electronmicroscopy showed that biomembranes were the main target, but nuclear damage was also de- monstrable. Addition of antioxidants decreased phototoxic cell death, indicating the participation of reactive oxygen species. This model proved not only useful in screening procedures, but also seems suitable for investigating mechanisms of action. INTRODUCTION It is important to identify possible photosensitizing agents before introduction into clinical therapy. A number of different test systems, ranging from red blood cells, yeast, and tissue cultures to animal models have been used to evaluate potential phototoxicity. Screening procedures are endorsed by the European Community countries (1). So far no in vitro or in vivo method has been able to recognize all known photosensitizers, and it is generally recommended to investigate several test models. The use of different light sources with varying emission spectra is only one of the factors responsible for conflicting results. On the other hand, the absorption spectra of the chromophors may change due to metabolism and binding to other components. Humidity, heat, and immunologic factors are other aspects that influence the results of phototoxicity in in vitro and in viva testing. Sulphonamide was introduced into clinical practice in the 1930s (2), and photosensi- tivity reactions due to sulphonamides were recognized shortly thereafter (3-5). By 167
168 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS chance, blood sugar-lowering and diuretic properties of oral sulphonamides were dis- covered, which led to the development of the oral antidiabetic drugs and the thiazides and loop diuretics (6). Clinical reports shortly after the introduction of these drugs showed photosensitizing properties with the antidiabetic (7) and diuretic derivatives (8,9) as well. In the industrialized countries, 16% of the population above 65 years suffers from diabetes mellitus and receive treatment with diet and oral antidiabetics (10). Similar prescription rates are found with the diuretics: in 1980 22.5 billion daily dosages were prescribed worldwide (11). Despite the extensive use of these substances, so far only two oral antidiabetics and eight diuretics have been investigated in vitro for phototoxic properties. The aim of this work was, on the one hand, to investigate the usefulness of the NHIK 3025 cell line, a model established in porphyrin research (12), in screening for photo- toxicity and mechanism studies, and on the other hand, to evaluate the potential phototoxic effects to a number of sulphonamide-derived substances. MATERIAL AND METHODS IRRADIATION SOURCES A broad-band UV irradiator ("Bluelight 2000" apparatus, H6nle, Martinsried, FRG) emitting 290-680 nm (Figure 1) and four fluorescent UVA tubes (Model 3026, Pho- tophysics, London, UK) emitting 340-420 nm, with an emission maximum at 405 nrn (Figure 2), were used in these studies. The emission spectra of the lamps was provided by H6nle and Photophysics, respectively. UVA and UVB fluence rates and fluence doses were measured with an integrating instrument (Centra-UV, Osram, Munich, FRG) E rel. I 250 270 290 310 330 350 370 390 410 430 450 Figure 1. Emission spectrum of the "Bluelight 2000" apparatus (H6nle, Martinsried, Germany) (1) and the transmission through the tissue culture flasks and mylar plastic (2).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)