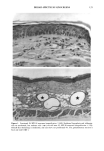
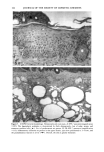
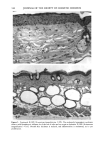
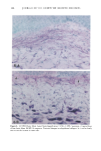
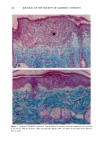
PHOTOTOXICITY TESTING 169 E rel. ^ i i i i i • I s I $ I I I I I t I I I • I • I I I I I I I I 350 375 400 425 450 475 500 Figure 2. Emission spectrum of the four fluorescent UVA tubes (Model 3026, Photophysics, London, UK). equipped with two filtered photodiodes with different spectral sensitivities. A part of the UVB portion of the "Bluelight 2000" apparatus was absorbed by the tissue culture flasks. To eliminate the remaining main part of the UVB, a sheet of mylar plastic with a thickness of 36 }xm was placed between the light source and the tissue culture flasks. The transmission of UV through the tissue culture flasks and mylar plastic was deter- mined with a spectrophotometer (UV/visible spectrophotometer, Perkin-Elmer, Oslo, Norway) (Figure 1). TEST SUBSTANCES The antidiabetic drugs chlorpropamide, glibenclamide, glipizide, gliquidone, glymi- dine, tolazamide, and tolbutamide, and the diuretics bemetizide, bendroflumethiazide, benzylhydrochlorothiazide, bumetanide, butizide, chlortalidone, furosemide, hydro- chlorothiazide, hydroflumethiazide, indapamide, piretanide, polythiazide, trichlorme- thiazide, and xipamide were dissolved in dimethylsulphoxide to a final concentration of 0.25 M. When 1-10 }xl of these solutions were added to 5 ml PBS buffer and incubated together with the cells, final test substance concentrations ranged from 0.05-0.5 mmol/l. PHOTOTOXICITY ASSAY NHIK 3025 cells, derived from a human carcinoma in situ of the cervix (13) were subcultured twice a week in Medium E2a (14) (split ratio 1:100) containing 20% human serum and 10% horse serum. The cells, 400 per flask (25 ml), were left at 37øC for 24 hours before addition of the test substance and irradiation. The cells were incubated together with the test substances for one hour. In the antioxidant experiments, ascorbic acid, alpha-tocopheol, beta-carotene, or ubiquinone (Q 10) were added to the test suspensions. Ascorbic acid, alpha-tocopherol, and beta-carotene were purchased from Sigma Chemical Company. Ubiquinone (Q 10)
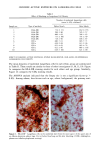
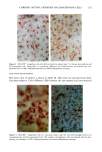
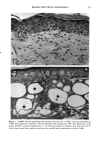
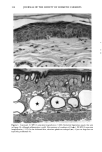
170 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS was kindly provided by Pharma Nord, Denmark. Ascorbic acid was dissolved in PBS (Dulbecco's phosphate-buffered saline) to a concentration of 0.5 mol/l, beta-carotene was dissolved in PBS buffer to a concentration of 0.025 mol/1, and alpha-tocopherol and ubiquinone were dissolved in DMSO to a concentration of 0.5 mol/1 and 0.25 mol/1, respectively. Of the antioxidant solutions, 5-20 p•l were added to 5 ml PBS buffer containing the test substance. The final concentration of the antioxidants added to the cell culture flask ranged from 0.025-2 mol/1. After incubation, the flasks were irradiated from above for 0-32 minutes (0-19.2 J/cm 2 UVA) with mylar-filtered irradiation from the "Bluelight 2000" apparatus or from below for 0-120 minutes (0-4 J/cm 2 UVA) with unfiltered irradiation from the UVA tubes. Control experiments with DMSO alone, DMSO and mylar-filtered light from the "Bluelight 2000" apparatus or UVA from the UVA tubes without photosensitizers, as well as irradiation with nonfiltered light from the "Bluelight 2000" apparatus, were performed. After irradiation 5 ml E2a medium with 30 per cent serum was added to the flasks, and the cells were incubated at 37øC for seven days. Then the cell colonies were fixed with absolute ethanol, stained with methylene blue, and counted. Cell survival was studied by measuring the colony-forming ability of the cells. Results are given as the median value of three experiments. For electronmicroscopic investigations, the test substance bemetizid in the 0.5 mM concentration was exposed to 2.5 J/cm 2 UVA from the "Bluelight 2000" apparatus. Five minutes after the end of irradiation, the cells were fixed with a mixture of 1% glutaraldehyde and 4% formaldehyde in 0.1 M cacodylate buffer, postfixed with cac- odylate-buffered 1% osmium tetroxide, and dehydrated in graded ethanols and propyl- ene oxide before being embedded in an Epon/Araldite mixture. Ultrathin sections were cut with diamond knives, floated onto 200-mesh copper grids covered by a supporting membrane, stained with uranyl acetate and lead citrate, and observed under a Philips CM-10 transmission electron microscope. RESULTS Phototoxic cell death was induced in the presence of the antidiabetic drugs glibencla- mide and gliquidone, and the diuretics bemetizide, bendroflumethiazide, benzylhydro- chlorothiazide, bumetanide, butizide, hydrochlorothiazide, hydroflumethiazide, piret- anide, polythiazide and trichlormethiazide after irradiation with the "Bluelight 2000" apparatus (Table I). Irradiation in the presence of the antidiabetics chlorpropamide, glipizide, glymidine, tolazamide, and tolbutamide and the diuretics chlortalidone, furosemide, indapamide, and xipamide did not induce a phototoxic reduction in the number of colony-forming cells. Irradiation with the UVA tubes induced phototoxic cell death in the presence of the antidiabetic glibenclamide and the diuretic bendroflume- thiazide, both in the concentration 0.5 mmol/1. Irradiation for one hour (2 J/cm 2 UVA) reduced the cell survival to about 1/40 in the presence of glibenclamide and to 1/10 in the presence of bendroflumethiazide. Two hours of irradiation (4 J/cm 2 UVA) com- pletely killed all the cells in the presence of both test substances. Addition of ascorbic acid and alpha-tocopherol reduced the phototoxic cell death in all the drugs investigated. The effects were dependent on the UV dose administered, the test substance concentration, and the antioxidant concentration. The inhibiting effects
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)