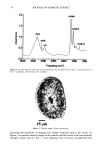
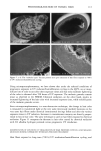
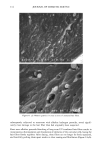
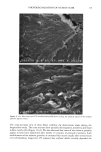
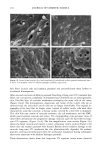
DIMETHICONE COPOLYOL 97 The data shows that the lower limit for surface tension for compounds of this type is 20 dynes/cm 2 (Table IV). This compares with typical values and is known to be caused by the low surface energy of the methyl groups in the flexible silicon backbone (1,6). The lack of significant differences in cmc is not very surprising since the hydrophobe- lipophile content is balanced by EMW considerations. The results can also be implied by the cloud point results, which do not vary much either. SPREADING Spreading area was studied relative to water on polyethylene films. Siloxane surfactants are well known for their wetting and spreading properties. These properties make them ideal for use in textiles, coatings, and agricultural adjuvants. For example, spreading and wetting properties are directly related to flow in coatings, spread-out areas in cosmetics formulations, and leaf coverage in pesticide formulations. Silicones typically spread better than conventional organic and fiuorocarbon surfactants. We measured the spreading properties for the various DMCs prepared. Our results, shown in Figure 1, indicate that the isomers studied spread slightly better than water. An optimum was observed at MD2*D2M , clearly outperforming the NP-9EO control. However, none of the samples can be considered superspreaders that, for example, would give a relative value of over 40. These materials are too hydrophilic, as shown by their high cloud point, and thus do not contain the needed subphases (or aggregates) that provide the necessary surfactant concentration gradient, and diffusion rate, in the droplet spreading front that drives the spreading. Literature states that the larger the molecule the slower the spread of the droplet front, and thus the slower the spreading (7-9). This is probably related to monomer size hindrance (molar volume) on packing at the sub- strate surface, possibly higher intrinsic viscosity. FOAMING The degree of polymerization seems to have an effect on foam height (Figure 2). Lower- molecular-weight material showed higher initial (flash) foam. However, the foam sta- bility was similar for both lower and higher molecular weight. EMULSIFICATION The results shown above for solubilization indicate that the lower-molecular-weight products will make good emulsifiers in an aqueous-based emulsion. This was found to Table IV Surface Tension and CMC 8 at CMC Designation CMC (mg/L) (dynes/cm 2) MD*DM 3 20 MD2*D2M 4 19 MD3*DsM 6 23 MD3*D7M 5 21 MD4*DsM 14 21
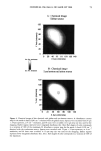
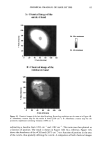
98 JOURNAL OF COSMETIC SCIENCE , •2' 1' o Figure 1. Spreading in polyester at 24øC and 0.1% w. Initial []After $ min. E 1• • 1, . o •7 2111 Molecular Weight Figure 2. Foaming at 1% w. be the case, as shown in Figure 3. The stability of the oil-in-water emulsion increased with lowering of the DMC molecular weight. WETTING We evaluated the DMC compounds using the Draves Wetting Test. Results are shown in Figure 4. The data show that there is a strong relationship between molecular weight (and structure) and wetting for DMCs. The lower-molecular-weight materials have faster wetting times. This finding is a trend also observed for other surfactants where shorter or branched structures provide faster wetting (10,11). The smaller molecule allows for more efficient packing efficiency and dynamics. The materials with lower molecular
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)