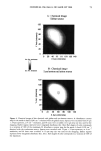
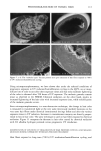
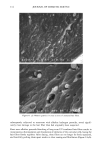
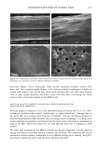
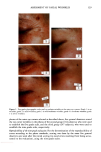
120 JOURNAL OF COSMETIC SCIENCE Figure 14. Longitudinal views of long-term UV-irradiated hair fibers and subsequent exposure for various times to 6% alkaline hydrogen peroxide: (a) no peroxide post-treatment, (b) 5 s, (c) 20 s, (d) 15 rain, (e) 15 rain (focused on dome of fiber), and (f) 60 rain of peroxide post-treatment. UV exposure in the QUV. Fibers exposed to continuous UV light at constant 10% hu- midity in the QUV do not show thinning and fusion of the cuticle cells (Figure 15a), as was observed in our earlier studies in which hair had been exposed to UV irradiation/ humidification (at 95% RH) cycling in the QUV. However, upon subsequent immer- sion in water, collapse and fusion of the surface cuticle cell occurs (Figure 15b), with a severity similar to that observed in hair fibers exposed to irradiation/humidification cycling in the QUV. This indicates that photodegradation had occurred at a low con- centration of water in the fiber structure, but that the degraded protein fractions (unlike in the swollen fiber), were not able to diffuse from the cuticle cell due to lack of adequate amounts of water to promote swelling. This thinning and fusion phenomenon can be easily explained. At very low relative
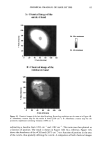
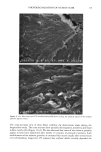
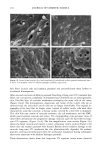
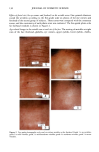
PHOTODEGRADATION OF HUMAN HAIR 121 Figure 15. Longitudinal views of hair fibers exposed to (a) 300 h of continuous UV irradiation at constant 10% RH, 50øC, and (b) subsequent 1-h immersion in lukewarm deionized water. humidities, below 25%, water molecules are principally bound to hydrophilic sites of the keratin fiber by hydrogen bonds, and the water is not mobile (13). As the humidity increases, additional water is sorbed, occupying the remaining adsorption sites associated with the protein. At high RH, when all the adsorption sites of the keratin are occupied, water molecules are no longer bound and are more mobile within the fiber. This facilitates diffusion of degraded, highly soluble, low-molecular-weight peptide fractions
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)