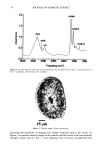
100 JOURNAL OF COSMETIC SCIENCE EYE IRRITATION Perhaps one of the most interesting properties evaluated in our study is the effect of molecular weight upon eye irritation. There have been many models proposed for the irritation properties of surfactants. We had thought that molecular weight might be one of the factors related to eye irritation since it would affect the diffusion of the material through the cell layers. We found that the higher the molecular weight the lower the ocular irritation (Figure 5). The results do in fact indicate that the irritation potential is related to molecular weight. The data was also quite surprising in that there is a very sharp drop in irritation as the molecular weight is increased, specifically until the molecular weight of the DMC reaches more than 1108. CORRELATION OF PROPERTIES The data presented here allows for the selection of cost-effective materials that have both good performance properties and possess low irritation potential. This is obtained when the data for ocular irritation is compared to that of other properties measured, as shown in Figure 6. For example, it allows one to predict that molecules having a molecular weight between 600 and about 1200 should be mild and effective wetting agents. CONCLUSION The proper selection of a dimethicone copolyol can result in a product that has a desirable combination of properties for various applications, such as personal care for- mulations. The work presented here indicates that there are molecules that are very [] 1 day 17 days 3O 25-- .•. 'r. 10-- 0 Figure 5. Draize primary ocular irritation (scale: moderately irritating 25.1-50 mildly minimally irritating 2.$-15, practically non-irritating 0.$-2.5, non-irritating 0-0.5).
DIMETHICONE COPOLYOL 101 40 .u 20• lO • .=• o 500 1000 1500 2000 2500 • Irritation Wetting .... O/W Stability - - - Non-polar Solubility Molecular Weight Figure 6. Correlation of primary ocular irritation and physical properties. efficient compromises when one desires a low irritation potential. In general, the trends and observations made include: ß Solubility in polar media seems to relate to the amount of polyoxyethylene group. ß Products with higher molecular weight had better dispersibility in nonpolar oils. ß The cloud point is related to the amount of polyoxyethylene group in the molecule and is rather independent of the silicone portion of the molecule. ß The molecules studied spread better than water but cannot be considered superspread- ers. ß The lower-molecular-weight materials have faster wetting times, higher initial foam, and are more stable than the emulsion of oil in water. ß The higher the molecular weight the lower the ocular irritation. REFERENCES (1) R. M. Hill, "Siloxane Surfactants," in Specialty Surfactants, I. D. B. Robb, Ed. (Blackie, London, 1997), p. 143. (2) I. Schlachter and G. Feldmann-Krane, in Novel Surfactants, K. Holmberg, Ed., Surfactant Science Series, Vol. 74 (Marcel Dekker, New York, 1998), p. 201. (3) R. M. Hill, Silicone Surfactants, Surfactant Science Series, Vol. 86 (Marcel Dekker, New York, 1999). (4) A. L. Smith, Ed., The Analytical Chemistry of Silicones, Chemical Analysis Series, Vol. 112 (John Wiley & Sons, New York, 1991). (5) J. H. Draize, "Dermal Toxicity," in Appraisal of the Saj•ty of Chemicals in Food, Drug and Cosmetics (The Association of Food and Drug Officials of the United States, 1954), pp. 49-51. (6) S.C. Vick, Soap/Cosmet./Chem. Spec., 36 (May 1984). (7) K. P. Ananthapadmanabham, E. E. Goddard, and P. Chandar, Colloids Surfaces, 44, 281 (1990). (8) T. Svitova, H. Hoffmann, and R. M. Hill, Lungmuir, 12, 1712 (1996). (9) T. Stoebe, Z. Lin, R. M. Hill, M.D. Ward, and H. T. Davis, Lungmuir, 12, 337 (1996). (10) N.E. Prieto, W. Lilienthal, and P. L. Tortorici,J. Am. Oil Chem. Soc., 73, 9 (1996). (11) M.J. Rosen, J. Am. Oil Chem. Soc., 49, 293 (1996).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)