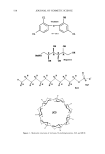
INCREASED AQUEOUS SOLUBILITY OF TRICLOSAN 539 water solubility 10 -6 g/m1-1. This lipophilic, anionic compound is compatible with many raw materials and has several dermatological uses (2). Among others, it has beneficial effects on atopic dermatitis (3,4), it reduces eczema (2) and plaque (5,6), and it can eliminate the irritant effects of sodium lauryl sulfate on the skin or buccal/lingual surfaces (6). All these applications make this antibacterial agent very useful for skin care formulations, particularly for surfactant-based hand soaps, hand disinfectants, mouth rinses, and surface cleaners (2). In two reports published in Nature, researchers showed that triclosan, far from being a generalized antimicrobial, works more like an antibiotic (7,8). Recently it was also demonstrated that triclosan kills the parasites responsible for malaria and toxoplasmosis, even at very low concentrations (9,10). Although triclosan is extensively used in cosmetic products and household chemicals, poor aqueous solubility does limit its wider application (11). Usually the poor solubility of lipophilic compounds is increased by solubilization, complexation, or salt formation (12). Savage (1) published a table listing the solubility of triclosan in commonly used solvents. To increase the solubility of triclosan, the compound has also been processed and complexed with various cyclodextrins including [3-cyclodextrin ([3CD) (11,13), •/-cyclodextrin (•/CD) (14), 2-hydropropyl-[3-cyclodextrin (HP[3CD) (15), and sulfobu- tyl ether [3-cyclodextrin (SB[3CD) (13). In all these studies the anionic triclosan/cationic SB[3CD complex increased the solu- bility of triclosan the most. Triclosan has also been converted to salts such as triclosan monophosphate to increase its solubility (16). In addition to these reports, no other detailed studies describing the enhancement of the aqueous solubility of triclosan have been found in the literature. In this study we report the effect of various additives on the solubility of triclosan in water. These solubilizing agents included ethanolamine, diethanolamine, triethanol- amine, glycine, L-arginine, N-methylglucamine (meglumine), [3CD, •/CD, HP[3CD, sodium benzoate, sodium methyl 4-hydroxybenzoate, and sodium lauryl sulfate (SLS). In addition, the antimicrobial activity of combinations of triclosan and those solubilizers that increased its solubility was compared to the antimicrobial activity of triclosan and the solubilizing agent alone. MATERIALS AND METHODS MATERIALS Triclosan (Irgasan DP 300, Ciba Specialty Chemicals, Basel, Switzerland) was obtained from Adcock Ingram, Ltd. (Krugersdorp, South Africa). Ethanolamine, diethanolamine, triethanolamine, glycine, L-arginine, N-methylglucamine, [3CD, •/CD, sodium benzo- ate, sodium methyl 4-hydroxybenzoate, and SLS were obtained from Sigma Chemical Company (St. Louis, MO). HP[3CD was obtained from Janssen Biotech (Brussels, Bel- gium). All other solvents and chemicals were analytical grade and were used as received. SOLUBILITY MEASUREMENTS The solubility of triclosan was determined in distilled deionized water and in a 0.1 M phosphate buffer, pH 7.4 (8.62 grams of sodium phosphate dibasic and 5.42 grams of sodium phosphate monobasic in one liter of Milli-Q grade water) containing increasing concentrations ranging from 0-0.2 M of [3CD, •/CD, HP[3CD, and sodium lauryl sulfate, and 0-1.0 M of sodium benzoate, ethanolamine, N-methylglucamine, D-(+)-
540 JOURNAL OF COSMETIC SCIENCE glucosamine, diethanolamine, triethanolamine, glycine, L-arginine, and methyl 4-hy- droxybenzoate sodium. Triclosan sufficient to ensure saturation was suspended in 10 ml of water or buffer containing increasing amounts of the solubilizers. Duplicate samples were rotated end to end in test tubes with screw caps at 30øC for 48 hours to reach equilibrium. The solubility was measured at 30øC because at this temperature it was easier to control temperature fluctuations during testing and sampling. Suspensions were passed through a 0.45-}•m filter (Osmonics, Minnetonka, MN), and the amount of triclosan dissolved in the filtered solutions was determined by HPLC. HIGH-PERFORMANCE LIQUID CHROMATOGRAPHY (HPLC) The HPLC method used in this study complied with specifications for precision, accu- racy, selectivity, linearity, and ruggedness as required by the USP XXIV (16). The following reagents and equipment were used: a Hewlett Packard ! 050 high-performance liquid chromotographer (Agilent Technologies, Palo Alto, CA), equipped with a vari- able wavelength UV detector, pump, injection device, and computerized data analysis system, and a Luna C•8 column (2 •m, 150 x 4.6 mm Phenomenex, Torrance, CA), controlled at +20øC. The mobile phase was a mixture of methanol:water (85:15) con- taining 0.1% H3PO 4. The flow rate was 1.0 ml/min -•, the injection volume was 10 }•1, and the UV detection was at 210 nm. A triclosan calibration curve was prepared from a series of diluted triclosan solutions that were prepared by dissolving 10 mg of triclosan, accurately weighed, in 100 ml of methanol with the aid of an ultrasonic bath. From this solution, several dilutions ranging from 10 }•g/ml -• to 250 }•g/ml -• were prepared. A calibration curve of the area under the curve (AUC) versus concentration was linear [y = 70875x + 94.649 (R 2 = 0.9998)] and was used to determine the concentration of triclosan in unknown solutions. Examples of chromatograms are shown in Figure 2. ANTIMICROBIOLOGICAL ACTIVITY Samples were sent to the SABS (South African Bureau of Standards, Pretoria, South Africa) where zone inhibition tests were performed on the samples (SABS method 730: Antibacterial Efficacy of Solid and Semi-Solid Antiseptics). Organisms tested against were Escherichia co/i, Pseudomonas aeruginosa, Staphy/ococcus aereus, Aspergillus niger, and Candida a/bicans. The following method was used: First, the triclosan powder was tested. Then the solubilizers in water and in buffer without triclosan were tested to determine the antimicrobial activity of each of the solubilizers alone. Afterwards, solutions con- taining the highest possible common concentration of triclosan that could be dissolved in all the solubilizer solutions were tested for antimicrobial activity. For example: A saturated solution of triclosan in an aqueous 1.0 M N-methylglucamine solution was prepared and the concentration of triclosan determined by HPLC. This was repeated for all the solubilizers at various concentrations. Based on these results, the highest common concentration that could be dissolved in each of the solubilizer solutions was determined. Solutions containing this concentration of triclosan, the equivalent amounts of the solubilizer in water, and the buffer at pH 7.4 were prepared and tested for antimicrobial activity. The antimicrobial activity of solutions containing only the solubilizers at the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)