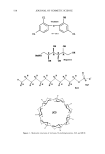
INCREASED AQUEOUS SOLUBILITY OF TRICLOSAN 541 Triclosan + Sodium Benzoate Triclosan + N-methylglucamin _,• Triclosa• ! ! 2 4 6 Time (rain) Figure 2. HPLC chromatograms of triclosan and triclosan solubilizer mixtures. desired concentrations was also tested. The aim of these tests was to determine if the antimicrobial activity of triclosan was influenced by the solubilizers. ZONE INHIBITION TEST (SABS SM 730:1975): ANTIBACTERIAL EFFICACY OF SOLID AND SEMISOLID ANTISEPTICS This test determined the ability of the triclosan solutions to inhibit the growth of Escherichia co/i, Pseudomonas aeruginosa, Staphylococcus aereus, Aspergillus niger, and Candida albicans. Sterile molten TS agar was prepared and allowed to cool to 45øC. While the agar was left to cool, 0. ! ml of solutions containing the respective microorganisms were piperred into sterile petri dishes. The agar was poured into the petri dishes and swirled to mix the agar and the microorganisms. The plates were left to cool and set, and then the agar was incubated for two hours at 37øC. A cork bore was used to make holes in the agar in the middle of each petri dish. The bottom of the holes was sealed with molten agar to stop diffusion of the liquid test products underneath the agar. Approximately 0. ! ml of the test solution prepared as described in the previous paragraph was poured into the holes, and then the plates were incubated for 48 hours at 37øC. This test measures the ability of triclosan solutions to diffuse into the agar and kill the microorganisms. After incubation, Vernier caliper was used to measure the angular radius of the zone that formed around the hole. STATISTICAL ANALYSIS All calculations were performed in Microsoft Excel (Microsoft, Seattle, WA). Multivari- ate analysis of variance (MANOVA), including apost hoc comparison using the Newman-
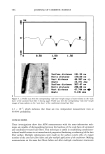
542 JOURNAL OF COSMETIC SCIENCE Keuls test, was performed on the mean inhibition zones and mean solubility values to identify significant differences in solubility and antibacterial activity (Statitica 5.1, Statsoft Inc., Tulsa, OK). P-values of less than 0.05 indicated significant differences in solubility. RESULTS AND DISCUSSION Triclosan (Figure 1) belongs to a class of compounds known as hydroxydiphenyl ethers. It is an anionic, lipophilic compound that is very poorly soluble in water. In this study, the solubility in water and pH 7.4 phosphate buffer was determined to be 0.002 mg/ml- • and 0.004 mg/ml- • at 30øC. In Figures 3 and 4 solubility profiles of triclosan in combination with increasing concentrations of three cyclodextrins and SLS are given. Previous researchers have reported the effect of cyclodextrins on the solubility of triclo- san (11,13-15). The results are given here as a basis for comparison, to evaluate the effect of other solubilizing agents on the solubility of triclosan. COMPLEXATION WITH CYCLODEXTRINS [•CD and HPI•CD both significantly increased the solubility of triclosan in water and the phosphate buffer (2000- to 4000-fold, Table I). Both cyclodextrins are soluble in water, but HPI•CD is more soluble because substitution of the hydroxyl groups of the [3CD disrupts the network of hydrogen bonding around the rim of the [•CD. As a result of disruption of the hydrogen-bonding network, the hydroxyl groups interact much more strongly with water, resulting in increased solubility compared to [3CD. Each 14 12 10 0.00 0.25 -•- Beta-CD J -e- Gamma-CD J -e-HPBCD • T 0.05 0.10 0.15 0.20 Concentration (M) Figure 3. Solubility of triclosan (mg/ml-•) at increasing concentrations of several cyclodextrins and SLS in water. The data points and error bars represent the mean and standard deviations of two replicates.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)