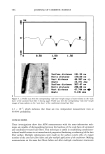
INCREASED AQUEOUS SOLUBILITY OF TRICLOSAN 545 14 12 10 -•- Meglumine -e- Ethanolamine -e- Diethanolamine + Triethanolamine 0.0 0.2 0.4 0.6 0.8 1.0 1.2 Concentration (M) Figure 5. Solubility of triclosan (mg/m1-1) at increasing concentrations of several amino alcohols in water. The data points and error bars represent the mean and standard deviations of two replicates. 2O 16 A Meglumine -e- Ethanolamine -e- Diethanolamine T • "r T 'r I ! 0.0 0.2 0.4 0.6 0.8 1.0 1.2 Concentration (M) Figure 6. Solubility of triclosan (mg/m1-1) at increasing concentrations of several amino alcohols in pH 7.4 phosphate buffer. The data points and error bars represent the mean and standard deviations of two replicates. of drugs such as phenobarbital (17). Such a triclosan/amino alcohol complex will form hydrogen bonds with water molecules through the hydrophilic hydroxyl groups of the alcohol moiety to increase the water solubility. In this study, it was not possible to
546 JOURNAL OF COSMETIC SCIENCE isolate these association complexes from solution. This provides some indication of the instability of these complexes. Although slight increases in the pH of the media resulted from an increase in amino alcohol concentration, solubilization of triclosan by this increase in pH was not signifi- cant because even higher concentrations of triclosan were achieved in solutions buffered at pH 7.4 (Table I). At a concentration of 1.0 M N-methylglucamine (Table I), the solubility of triclosan in the phosphate-buffered solution was =50% higher than in water. This phenomenon could be attributed to an increase in the solvent activity caused by the buffer salts in combination with N-methylglucamine (salting-in), leading to an increase in the solubility of the complexes (19). The same media-dependent increase in solubility was not observed with the other amino alcohols but was observed for L- arginine. Figures 7 and 8 represent solubility profiles of triclosan in combination with two highly water-soluble amino acids and two water-soluble preservatives. The two preservatives, sodium benzoate and sodium methyl 4-hydroxybenzoate, increased the solubility of triclosan, but this increase was not as significant as that obtained with N- methylglucamine, [3CD, HP[3CD, and SLS. However, the combination of these preser- vatives with triclosan could prove useful for the formulation of products where preser- vative combinations might reduce the chance of bacterial resistance. The acidic amino acid glycine (Figure 7) did not increase the solubility of triclosan, but the strongly alkaline L-arginine did increase the solubility of triclosan in aqueous solutions. The mechanism whereby it increases the solubility of triclosan is probably similar to that of N-methylglucamine because of structural similarities, especially the amine groups (10). 14 12 10 •, Glycine -e- L-Arginine - Na-Benzoate + Na-Methylparaben 4. 4, .I. .I. 0.0 0.2 0.4 0.6 0.8 1.0 1.2 Concentration (M) Figure 7. Solubility of triclosan (mg/ml -]) at increasing concentrations of two amino acids and other preservatives in water. The data points and error bars represent the mean and standard deviations of two replicates.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)