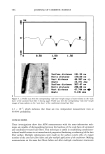
INCREASED AQUEOUS SOLUBILITY OF TRICLOSAN 547 2O -•- Glycine 16 -e- L-Arginine ..• ß Na-Benzoate = 8 0.0 0.2 0.4 0.6 0.8 1.0 1.2 Concentration (M) Figure 8. Solubility of triclosan (mg/ml -•) at increasing concentrations of two amino acids and other preservatives in pH 7.4 phosphate buffer. The data points and error bars represent the mean and standard deviations of two replicates. There was no significant difference in the solubility profiles (Figure 7 and 8) of triclosan combined with N-methylglucamine or L-arginine in both water and the buffer. Maxi- mum solubilities achieved at corresponding concentrations of the solubilizers were also not significantly different (Table I). EFFECT OF SOLUBILIZERS ON THE ANTIMICROBIAL ACTIVITY OF TRICLOSAN Triclosan possesses bacteriostatic activity at low concentrations when tested against most gram-negative as well as gram-positive bacteria by the agar incorporation method, a notable exception being Pseudomonas (1). Since the solubilizers increased the solubility of triclosan in water, it is important to know if these solubilizers influenced the antimi- crobiological activity of triclosan. Therefore, the antimicrobial activity of the five solu- bilizers that improved the solubility of triclosan in water the most, N-methylglucamine, L-arginine, ethanolamine, SLS, and [3CD, were tested alone and in combination with triclosan against Escherichia co/i, Pseudomonas aeruginosa, Staphylococcus aereus, Aspergillus niger, and Candida albicans. The size of growth inhibition zones listed in Table II show that the solubilizer with the best bacteriostatic activity was SLS against A. niger. However, this compound did not inhibit the growth of E. co/i or P. aeruginosa. Both N-methylglucamine and ethanolamine significantly inhibited the growth of all the organisms tested, while [3CD showed no zone inhibition at all. Triclosan inhibited the growth of all the organisms tested except P. aeruginosa. Statistically there were no significant differences between the growth inhibition zones
548 JOURNAL OF COSMETIC SCIENCE Table II Antimicrobial Activity, Measured by Zone Inhibition, of Triclosan and Solubilizing Agents Diameter of inhibition zone (mm) Compound E. coli P. aeruginosa S. aureus A. niger C. albicans Triclosan powder 18.8 No zone 19.5 17.4 14.5 N-methylglucamine 15.9 19.3 14.3 25.5 20.9 L-arginine No zone 14.7 No zone 24.6 20.1 Ethanolamine 23.9 24.6 12.9 27.8 18.4 SLS No zone No zone 18.2 28.6 15.1 [3CD No zone No zone No zone No zone No zone for mixtures of triclosan and the solubilizing agents prepared in water or the pH 7.4 phosphate buffer. In Table III the results for the phosphate buffer solutions are listed. The diameters of the zones of inhibition increased when a constant concentration of 6.0 mg/ml -• of triclosan was mixed with all the solubilizers. This showed that the bacte- riostatic efficacy of triclosan increased when combined with the solubilizers. Whether this was because of the bacteriostatic efficacy of the solubilizer, or a synergistic effect that occurred, is unknown. The results listed in Table III show that, on average, the combined solutions doubled the diameters of the inhibition zones. From these results, it is also clear that N- methylglucamine, L-arginine, and ethanolamine in combination with triclosan were the most effective antimicrobial combinations. Although not as effective as the other solu- bilizers, both [3CD and SLS also enhanced the growth inhibition of most of the organ- isms tested when combined with triclosan. The difference between the antimicrobial activity of solutions containing the complexing agents, N-methylglucamine, L-arginine, and ethanolamine, and the compounds that encapsulate the triclosan, [3CD and SLS, was significant. This difference could be the result of partial inactivation of the triclosan by miceliar entrapping on inclusion because these processes reduce the amount of free triclosan available in solution. Overall, triclosan/solubilizer combinations were most effective against A. niger and least effective against P. aeruginosa. CONCLUSIONS A solubilization study for triclosan in water and pH 7.4 phosphate buffer at 30 ø C Table III Antimicrobial Activity, Measured by Zone Inhibition, of Solutions Prepared in pH 7.4 Phosphate Buffer Containing Triclosan and Solubilizing Agents Diameter of inhibition zone (mm) Compound E. coli P. aeruginosa S. aureus A. niger C. albicans N-methylglucamine 34.2 17.4 32.7 42.4 29.4 L-arginine 34.9 19.1 34.1 44.2 31.4 Ethanolamine 35.0 23.5 31.5 42.5 31.2 SLS 23.5 12.4 21.0 30.4 23.2 •CD 30.5 No zone 23.1 27.8 27.2
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)