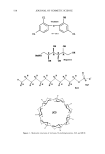
INCREASED AQUEOUS SOLUBILITY OF TRICLOSAN 549 showed the following compounds as potential solubilizers of the drug. They are in order of their solubilizing performance: N-methylglucamine -- L-arginine SLS [3CD -- HP[3CD ethanolamine sodium benzoate sodium methyl 4-hydroxybenzoate triethanolamine -- diethanolamine. By using these solubilizers, the solubility of triclo- san was increased 80- to 6000-fold. The formation of either salts or complexes is postulated as a possible mechanism for the increase in the solubility of triclosan by these compounds. In addition, the combination of triclosan with some of these solubilizers increased the antimicrobial potency of triclosan. The mechanism for this increase in activity is unknown and needs further investigation. However, complexing and salt- forming agents improved the activity of triclosan significantly more than compounds that solubilized triclosan by miceliar or molecular inclusion. REFERENCES (1) (2) (3) (4) (5) (6) (7) (8) (9) (10) (11) (12) (13) (14) (15) (16) (17) C. A. Savage, A new bacteriostat for skin care products, Drug Cosmet. Ind., 109, 36-38, 163 (1971). H. P. Nissan and D. Ochs, Triclosan: An antimicrobial active ingredient with anti-inflammatory activity, Cosmet. Toiletr., 113, 61-64 (1998). V. Kjaerheim, P. Barkvoll, S.M. Waaler, and G. Roella, Triclosan inhibits histamine-induced in- flammation in human skin, J Clin. Periodont., 22, 423-426 (1995). R. D. Jones, H. B. Jampani, J. L. Newman, and A. S. Lee, Triclosan: A review of effectiveness and safety in health care settings, Am. J. Infect. Control., 28, 184-196 (2000). S. M. Waaler, G. Roella, K. K. Skjoerland, and B. Oegaard, Effects of oral rinsing with triclosan and sodium lauryl sulphate on dental plaque formation: A pilot study, Scand. J. Dent. Res., 101, 192-195 (1993). V. Kjaerheim, S. M. Waaler, and A. Kalvik, Experiments with two-phase plaque-inhibiting mouth- rinses, Eur. J. Oral Sci., 103, 179-181 (1995). L. M. McMurry, M. Oethinger, and S. B. Levy, Triclosan targets lipid synthesis, Nature, 394, 531-532 (1998). C. W. Levy, A. Roujeinikova, S. Sedelnikova, P. J. Baker, A. R. Stuitje, A. R. Slabas, D. W. Rice, and J. B. Rafferty, Molecular basis of triclosan activity, Nature, 398, 383-384 (1999). S. Namita and A. Surolia, Triclosan offers protection against blood stages of malaria by inhibiting enol-ACP reductase of Plasmodiumfalciparum, Nat. Med., 7, 167-173 (2001). R. Perozzo, M. Kuo, A. S. Sidhu, J. T. Valiyaveetti, R. Bittman, W. R. Jacobs, D. A. Fidock, and J. C. Sacchettini, Structural elucidation of the specificity of the antibacterial agent triclosan for malarial enoyl acyl carrier protein reductase, J. Biol. Chem., 277, 13106-13114 (2002). T. Loftsson, N. Leeves, N. Bjornsdottir, L. Duffy, and M. Masson, Effect ofcyclodextrins and polymers on triclosan availability and substantivity in toothpastes in vivo, J. Pharm. Sci., 88, 1254-1258 (1999). S. H. Yalkowsky, Ed., Techniques of Solubilization of Drugs (Marcel Dekker, New York, 1981), pp. 16-34, 91-134, 135-157. J. Lu, M. A. Hill, M. Hood, D. F. Greeson, J. R. Horton, P. E. Orndorff, A. S. Hemdon, and A. E. Tonelli, Formation of antibiotic, biodegradable polymers by processing with Irgasan DP 300R (tri- closan) and its inclusion compound with [3-cyclodextrin,J. Fiber Polym. Sci., 82, 300-309 (2001). P.M. Ashall, B. M. Kiernan, and N. R. Russell, Complexation of the bacteriostat triclosan the •-cy- clodextrin, Proceedings of the World Meeting on Pharmaceutics, Biopharmaceutics and Pharmaceutical Tech- nology, Budapest, May 9-11, 1995, pp. 605-606. T. Loftsson, N. Leeves, J. F. Sigurjonsdottir, H.H. Sigurosson, and M. Masson, Sustained drug delivery system based on a cationic polymer and an anionic drug/cyclodextrin complex, Pharmazie, 56, 746-747 (2001). The United States Pharmacopeia, XXIVth Revision (United States Pharmacopeial Convention, Rockville, MD, 2000). J. K. Lim, H. O. Thompson, and C. Piantadosi, Solubilization and stability of phenobarbital by some aminoalcohols, J. Pharm. Sci., 53, 1161-1165 (1964).
550 JOURNAL OF COSMETIC SCIENCE (18) M.M. de Villiers, W. Liebenberg, S. F. Malan, and J.J. Gerber, The dissolution and complexing properties of ibuprofen and ketoprofen when mixed with N-methylglucamine, Drug Dev. Ind. Pharm., 25,967-972 (1999). (19) A. A1-Maaieh and D. R. Flanagan, Salt effects on caffeine solubility, distribution, and self-association, J. Pharm. Sci., 91, 1000-1008 (2002). (20) K. R. Morris, M. G. Fakes, A. B. Thakur, A. W. Newman, A. K. Singh, J. J. Venit, C. J. Spagnuolo, and A. T. M. Serajuddin, An integrated approach to the selection of optimal salt form for a new drug candidate, Int. J. Pharm., 105, 209-217 (1994).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)