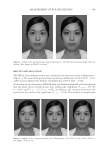
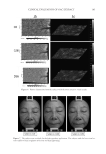
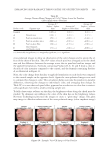
JOURNAL OF COSMETIC SCIENCE 146 production and mushroom tyrosinase activity in Melan-a cells. Moreover, UVB-induced histopathological dermal sclerosis and infl ammatory signs were signifi cantly attenuated in PCS-administered mice compared with UVB-exposed mice. Conclusions: Our results suggest that PCS prevents signs of aging, including those related to photoaging. These effects are associated with enhanced hyaluronan synthesis, as well as suppressed elastase, collagenase, MMP-1, and tyrosinase activities and melanin production. UVB-induced photoaging, such as histopatho- logical dermal sclerosis and infl ammatory signs, were effectively reduced on the addition of PCS. These re- sults also suggest that skin aging can be prevented and reduced by the antioxidant effects of PCS. INTRODUCTION Skin prevents the invasion of harmful components, serving as a protective barrier for the body. However, it is exposed to ionizing radiation, chemicals, toxins, and oxidative stressors from both endogenous and exogenous sources (1) these factors cause a loss of structural integrity and physiological function, ultimately leading to skin damage. Specifi cally, exposure of skin to ultraviolet (UV) radiation (UVA and UVB) is considered an important cause of skin aging. Consequently, skin can produce visible signs, such as fi ne wrinkles, irregular dryness, dyspigmentation, sallowness, deep furrows or severe atrophy, dehydration, skin roughness, telangiectasia, and premalignant lesions (2). UV irradiation can also induce the expression of matrix metalloproteinases (MMPs). MMPs, a large family of zinc-dependent endoproteases, have the ability to degrade all extracellular matrix (ECM) proteins (3). ECM degradation is exacerbated by aging and radiation, demonstrating a decrease in dermal collagen and an increase in MMP-1, which cleaves interstitial collagen (4). Furthermore, collagenase activity has been shown to be inhibited by tissue inhibitor of metalloproteases (TIMP) (5). Functional cosmetics have been used to prevent skin alterations, exerting potent antiag- ing properties (i.e., whitening, antiwrinkle, and moisturizing effects) (6). In particular, functional ingredients from natural sources represent very attractive materials due to their reactive oxygen species (ROS) scavenging potential and inhibition of the UV-induced signal transduction pathway (7,8). Thus, antioxidant substances may represent a promis- ing strategy for the prevention of skin aging. Pomegranates are consumed throughout the world for their benefi cial health effects. The pomegranate is comprised mostly of ellagic acid and other organic materials including fl avonoids and polyphenols (9–11). The pomegranate contains more antioxidants than both red wine and green tea. These characteristics provide protection against heart disease and cancer (12–17). In addition, the pomegranate possesses antiproliferative (18), anti- infl ammatory (19), and antitumorigenic functions (20). This evidence prompted us to examine the protective effects of pomegranate concentrated solution (PCS) on skin aging in vitro using human keratinocytes (HaCaT), mouse mela- nocytes (melanocytes), and normal human primary dermal fi broblast-neonatal cells (HDF-N fi broblasts). The antiwrinkle effects of PCS were determined through changes in hyaluronan synthesis in HaCaT cells. MMP-1 activity and procollagen synthesis were also examined in HDF-N cells due to the close correlations between wrinkle formation and loss of elasticity, collagenase, and MMP-1. Whitening effects were evaluated by a tyrosinase inhibition assay, and melanin formation was measured in Melan-a cells. In ad- dition, histopathological analysis on dorsal back skin tissue from UV-exposed mice was performed to explore the anti-photoaging effect of PCS in vivo.
THE WHITENING AND ANTI-WRINKLE EFFECT OF PONEGRANATE 147 METHODS AND MATERIALS CHEMICALS A solution containing pomegranate was kindly provided by Health-love Therapeutics (Anyang, South Korea), who purchased concentrated pomegranate juice from Asya Fruit Juice and Food Ind. Inc. (Ankara, Turkey). The preparation of pomegranate solution was as follows: according to the manufacturer’s method, briefl y, the fruit was washed and then the rind was removed. Next, the pomegranate juice and seeds were separated using Bucher press. The pomegranate juice was sterilized in a high-temperature condition. The sterilized pomegranate juice was depectinized by pectinase. Next, it was fi ltered and concentrated which contains ellagic acid (2.31 mg/g). All test materials were maintained at 4°C until use. The solution was diluted with distilled water to concentrations of 0.005–0.5%. CELL CULTURES HaCaT cells were purchased from the American Type Culture Collection (ATCC, Manassas, VA) and cultured according to the manufacturer’s protocols. Cells were maintained in Dulbecco’s modifi ed Eagle’s medium (DMEM Sigma-Aldrich, St. Louis, MO) with 10% fetal bovine serum (FBS Lonza, Walkersville, MD), 100 μg/ml streptomycin (Sigma- Aldrich), and 100 U/ml penicillin (Sigma-Aldrich). HDF-N cell lines were obtained from ATCC and were cultured in fi broblast basal medium (FBM Lonza) supplemented with 2% FBS, 0.1% insulin, 0.1% recombinant human fi broblast growth factor (rhFGF), and 0.1% gentamicin. Melan-a cells were provided by Dr. Dorothy and were cultured in RPMI 1640 medium supplemented with 10% heat-inactivated FBS, 50 μg/ml strepto- mycin, and 50 U/ml penicillin. Cells were cultured at 37°C in a fully humidifi ed atmo- sphere of 5% CO2 and were passaged approximately every other day. WST-1 CELL PROLIFERATION ASSAY HaCaT, HDF-N, and Melan-a cells were plated at a density of 5 × 104, 6 × 103, and 9 × 103 cells/well in a 96-well plate, respectively. The HaCaT, Melan-a, and HDF-N cells were exposed to various concentrations of PCS for 24 h and then cell proliferation reagent WST-1 (Roche, Mannheim, Germany) was added to each well and incubated in an incu- bator for 1 h. The absorbance of the wells at 450 nm was read using a microplate reader (TECAN, Männedorf, Switzerland). HYALURONAN ASSAY To determine the activity of hyaluronan synthesis, HaCaT cells were treated with 0.01%, 0.05%, and 0.1% PCS or N-acetyl-D-glucosamine (NAG) for 24 h, and then cells were trypsinized and counted for normalization. Hyaluronan concentration in the samples was quantifi ed using an enzyme-linked hyaluronan-binding protein sandwich assay (Cat no. DY3614 R&D Systems, Minneapolis, MN) based on the manufacturer’s methods (21).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)