JOURNAL OF COSMETIC SCIENCE 178 IN VIVO CLINICAL TEST Ethical considerations and declaration. This study was conducted in compliance with the ethical principles of the Offi ce of Ethics Review Committee for Research Involving Human Research Subjects, Health Science Group. The study was reviewed by the appro- priate ethics committees and approved for clinical studies. The validation number of the “Code of Approval” granted to this study is 079/2557. This study was conducted accord- ing to the ethical principles of the Helsinki Declaration (1964). Subjects. A total of 22 female subjects aged 45–61 years who had slight wrinkles or fi ne lines (crow’s feet) participated in the study. The subjects were recruited from a pool of volunteers at Dermscan Asia according to the Helsinki Declaration (1964) via a volunteer recruiter. They were instructed to systematically apply cream to the entire face twice daily. Whitening, moisturizing, and antiwrinkle measurements were obtained after 28 and 56 days of product application. Cutaneous hydration measurements. Cutaneous hydration was measured with a COURAGE & KHASAKA CM 825® Corneometer (Cologne, Germany). This instrument determines the humidity level of the most external cutaneous layers of the stratum corneum. The princi- ple of action of the Corneometer involves the modifi cation of the electrical capacities of the detector, which is designed as a condenser. The electrical capacity of the surface of the measurement tool head (which is in contact with the skin) varies according to the level of skin humidity. Changes in the dielectric constant due to skin surface hydration variations alter the capacitance of the probe. Skin color measurement. The Minolta Chroma Meter CR-321® is a colorimetric instrument that contains a xenon lamp as a light source as well as photodetectors, a microcomputer, and colored fi lters that closely match the CIE colorimetric Standard Observer curves (L*a*b* color system). The Chroma Meter converts colors perceived by humans into a digital code composed of three parameters: L*: for clarity (from dark to light) a*: for the green-to-red spectrum b*: for the blue-to-yellow spectrum a* and b* are the chrominance parameters and L* is a luminance parameter. These parameters enable close comparisons between two cutaneous zones that appear to be the same color. After the calibration phase, the measurements are performed directly on the skin using a pulsed Xenon light source as well as a dual beam system that measures the light transmitted (to correct slight deviations). Both parameters L* (luminance) and b* (cutaneous melanin yellow color) are assessed during a depig- menting product study. L* and b* are used to calculate the Individual Typological Angle (ITA°), which defi nes the skin pigmentation of a subject according to the following formula: ITA° = [Arc tan ((L*−50)/b*)] × 180/π The higher the ITA°, the lighter the skin. Thus, the higher the parameter L*, the lighter the skin the lower the parameter b*, the less yellow the skin.
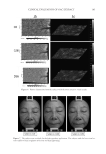
CLINICAL EVALUATION OF GAC EXTRACT 179 Variations (Δ) in the colorimetric parameters L* and b* as well as the ITA° were calcu- lated in arbitrary units according to the following formula: Δ = Ti−T0 where Ti is the value of the colorimetric parameter at the treated zone on days 28 and 56 and T0 is the value of the colorimetric parameter at the treated zone on day 0. Skin roughness measurement. PRIMOS (Phaseshift Rapid In Vivo Measurement of the Skin), Berlin, Germany (20) was used to determine surface roughness. This device uses micro 3D to obtain a three-dimensional (3D) image of the skin to measure facial wrinkles (e.g., crow’s feet, forehead wrinkles, glabella, and nasolabia) and skin roughness. The device also microprofi les the skin surface to identify wounds, scars, acne, allergies, and wheals. The entire face or parts of the body can be assessed in preparation for plastic surgery, maxillofacial surgery, and other treatments. PRIMOS is a reliable and objective tool for evaluating surface roughness of normal-appearing skin and burn scars (21). PRIMOS has been used to measure skin appearance parameters such as wrinkles, scars, and roughness (21). A study of the cutaneous relief parameters (length, depth, and wrin- kle area in μm) was completed using PRIMOSlite® (GFMesstechnik GmbH, Berlin, Germany) with an alysis software. The PRIMOSlite device represents recent develop- ments in 3D skin measurement. The associated technique involves the direct 3D analysis of wrinkles at a lateral angle of the eye in vivo without the use of replicas. This method involves the projection of a pattern with parallel fringes onto the measurement object and observation of the pattern from an angle other than the angle of projection. The projected pattern with parallel fringes is distorted by the unevenness of the object’s surface. An analysis of the distorted pattern generates 3D measurements. The acquisition software enables 2D and 3D measurements and obtains cutaneous relief parameters on radii dis- tributed in a star pattern over the zone of interest. An automatic repositioning system enables the precise reidentifi cation of the measurement zone illustrated by Figure 1. Figure 1. Radii distributed in a star pattern over the zone of interest. PRIMOSlite also creates a phase image from images with interference fringe projections. This image then facilitates the determination of the height of each point. The acquisition software enables 2D and 3D measurements to be obtained and to determine the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)