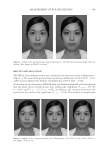
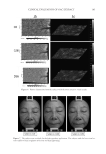
JOURNAL OF COSMETIC SCIENCE 180 The following parameters were assessed for each profi le (the analysis was based on the average of 32 profi les): • Ra, the average roughness (in μm): defi ned similar to the ratio between the surface integrated around the mean value of the profi le. A decrease in Ra indicates a smoothing effect. • Rz, the average relief (in μm): the average value of all of the maxima (between peaks and troughs) of the profi le. A decrease in Rz indicates an antiwrinkle effect. • Rt, the amplitude (in μm): average value of the fi ve highest maxima (between peaks and troughs) of the profi le. A decrease in Rt indicates an antiwrinkle effect. Illustrations with the VISIA® system. Product effectiveness was evaluated using full-face photography via VISIA® from CANFIELD® imaging systems (Fairfi eld, NJ). VISIA® allows pictures to be acquired under varying illumination types at a very rapid capture rate. Repositioning occurs on a data processing screen using an overlay visualization of the images at each acquisition. A series of photos acquired with multispectral imaging and analysis [white light, UV, or polarized light (parallel or crossed)] enables the capture of visual information relevant to complexion health and appearance in Figure 3. Figure 2. The average roughness is represented by Ra (in μm), the average relief is represented by Rz (in μm), and the average value of all of the maxima (between the peaks and troughs) is represented by Rt. Figure 3. A series of example photos acquired with multispectral imaging and analysis using VISIA® [white light, UV, or polarized light (parallel or crossed)]. parameters of the cutaneous relief for 32 radii distributed in a star-like pattern in the zone of interest. An automatic repositioning system enables the precise reidentifi cation of the zone of measurement. Digital stripes are spotted onto the skin. The refl ected light is ab- sorbed by a chip with a high-resolution camera and height variations are recorded to produce a 3D image in Figure 2.
CLINICAL EVALUATION OF GAC EXTRACT 181 Safety and irritation. The product’s safety was assessed for potential adverse events. The acute skin tolerance of the cosmetic product was evaluated on 22 adult subjects via ap- plication under an occlusive patch over a 24- or 48-h period 2 samples of 30 ml or 30 g were used for each product. One product was assessed using “occlusive” or “semiocclusive” patch test types. The clinical examination of the scapular area was under dermatological control. Reactions were defi ned using the following criteria: • Erythema • Edema • Cutaneous dryness • Vesicles Table I shows the acute cutaneous tolerance of the product in 22 healthy subjects during a 48 h occlusive single patch test. Inclusion and exclusion criteria were assessed using the Mean Irritation Index (MII) (22–24). MII = Total cutaneous reactions score (erythema ± edema)/Number of subjects Table I Irritation Evaluation MII Product classifi cation MII ≤ 0.20 Non irritating 0.20 ≤ MII 0.50 Slightly irritating 0.50 ≤ MII 1 Moderately irritating MII ≥ 1 Irritating MII product classifi cation involves the following parameters: nonirritating (MII ≤ 0.20), slightly irritating (0.20 ≤ MII 0.50), moderately irritating (0.50 ≤ MII 1), and irri- tating (MII ≥ 1). No irritation (MII values of 0.00). Statistical analysis. Data are expressed as the mean ± standard deviation (S.D.) of three replicates. One-way analysis of variance (ANOVA) and Duncan’s new multiple range test were used to determine differences between means. The p values 0.05 were regarded as signifi cant. RESULTS AND DISCUSSION IN VITRO BIOLOGICAL TEST Antioxidant activities. The antioxidant activity of Gac extract was measured and compared with those of vitamins C and E. Gac extract exhibited the highest antioxidant activity in the DPPH, ABTS, and FRAP assays, as shown in Figure 4.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)