36 JOURNAL OF COSMETIC SCIENCE the origin, it usually contains resin and balsam (55%), honey (30%), aromatic oils (10%), and pollen (5%) (1). Flavonoids, as the major ingredients of propolis, exhibit antimicrobial, antiulcer, and wound healing effects. Propolis flavonoids (PFs) have also demonstrated antitumor, antibacterial, antifungal, antiparasitic, antioxidant, anti-inflammatory, and anticaries properties (2). Based on the wavelengths, ultraviolet (UV) rays, which are radiated by the sun, are divided into three categories of UVA (320–400 nm), UVB (290–320 nm), and UVC (290–320 nm). In contrast to UVC, which is absorbed by the ozone layer of the atmosphere, UVA and UVB can reach to the surface of earth and, therefore, have the potential for development of a wide range of skin diseases, from mild sunburn and light skin inflammations to severe malignancies (3). UVB radiations can penetrate the skin to a depth of 160–180 μm and cause wrinkles, scaling, dryness, dilation of blood vessels, and collagen loss as well as increase the risk of skin cancer and melanoma (4,5). UVA radiation can penetrate deeper into the epidermis and dermis and produce reactive oxygen species that can pose damaging effects on proteins, lipids, and DNA structures (6). Therefore, in recent decades, researches for development of a sun protection formulation have been intensified (7). Either two mechanisms of chemical (i.e., absorption of the radiation) or physical (i.e., blocking of the radiation) are involved in sun protection effects of sunscreens (8). Their ability to protect the skin is explained by sun protection factor (SPF) unit (9). This term indicates the degree of skin protection against scorching rays of the sun. However, the skin permeation of the common synthetic sunscreen components such as oxybenzone, avobenzone ecamsule, and octocrylene may pose serious side effects, including allergic reactions, DNA damages, antiandrogenic effects, and development of cancer and other health issues (10–12). Therefore, many researches are focused to find a natural sunscreen that is expected to have fewer side effects (13). Due to protective effects of propolis on the skin, it can be considered as a good candidate for incorporation in skin-care cosmeceuticals (e.g., sunscreen) (14). Moreover, propolis exhibits potent anti-inflammatory effects, and therefore it is suitable for treatment of sunburns and other radiation-dependent skin problems (15). Simultaneously, by preserving the collagen contents of the skin, it can act as an anti-aging product (16). Furthermore, since propolis contains various flavonoids and antioxidants, it can protect the skin from UV rays (17). The sun protection effects of the propolis are mainly attributed to the presence of PFs that can demonstrate the potential for direct absorption of UV radiations (4). In addition, due to exertion of antioxidant properties, these flavonoids can eliminate UV-induced ROS and exhibit skin protection effects against sunlight. However, due to poor skin penetration, application of propolis for skin protection is limited (18). It is suggested that the issue can be resolved by development of an efficient nanoparticulate drug delivery system incorporating PFs (19–21). Poly-lactide-co-glycolide (PLGA), due to its biocompatibility, biodegradability, and low toxicity, is one of the widely studied polymers in fabrication of drug delivery systems (22). Moreover, by controlling the particle size and surface charge, their cargo can be released slowly over the period of several days (23). The aim of this study was to develop a sunscreen formulation containing PF-encapsulated PLGA nanoparticles. For this purpose, an optimized formulation of nanoparticles that exhibited proper physicochemical characteristics including particle size, polydispersity index (PdI), zeta potential, entrapment efficiency (EE%), and drug release behavior was achieved. The nanoparticles were incorporated to a sunscreen formulation and their sun protection effect was determined using in vitro method.
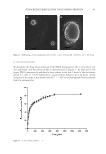
37 SUNSCREEN FORMULATION CONTAINING PROPOLIS MATERIALS AND METHODS MATERIALS Raw propolis samples were gifted from a local commercial beekeeper (Sabalan, Iran). Collected propolis was kept in a dry place and stored at 4°C for more studies. Galangin PLGA, polyvinyl alcohol, and cellulose membrane dialyzing tube (molecular weight cut off 12,000 g/mole) were all purchased from Sigma-Aldrich™ (St. Louis, United States). Aluminum chloride, dichloromethane, ethanol (99.7% v/v), potassium dihydrogen phosphate (KH 2 PO 4 ), dipotassium hydrogen phosphate (K 2 HPO 4 ), sodium hydroxide (NaOH), and isopropyl myristate were all provided by Merk-Millipore™ (Darmstadt, Germany). Cetomacrogol 1000® (polyethylene glycol hexadecyl ether) (Croda™ International, Snaith, United Kingdom) was provided as the gift from Croda™ International (Snaith, United Kingdom). Liquid paraffin and cetostearyl alcohol were purchased from Mojalal Co. (Tehran, Iran). Freshly prepared, deionized double distilled water was provided by using Mili-Q® ultra-pure water purification system (Merck Millipore™, Burlington, MA, United States). All other chemicals were of pharmaceutical grade and were used as received. PREPARATION OF PF EXTRACT The extraction procedure was performed according to previous studies with some modification (24,25). Briefly, 1 g of the propolis sample was added to 50 mL of ethanol and the mixture was stirred at room temperature for 72 hours. To avoid solvent evaporation, the container was covered by Parafilm® (Bernis Company, Inc., Neenah, WI, United States). To obtain a transparent solution, the ethanolic extract was then filtered through a Whatman® filter paper (Tisch Scientific, Cleves. OH, United States) using a Büchner funnel. The obtained extract was stored at 4°C for further experiments. The standardization of the extract was performed using galangin as the reference for quantification of total flavones and flavonoids (26). The spectrophotometric assay was performed based on the formation of a complex between aluminum (i.e., Al3+) and carbonyl- or hydroxyl- functional groups of the total flavones and flavanols (1). Briefly, a standard solution of galangin (100 µg/mL) was prepared by dissolving the precisely weighted amounts of galangin in ethanol. Then, a series of dilution were prepared from the stock solution to achieve concentrations of 50 μg/mL, 20 μg/mL, 10 μg/mL, 5 μg/mL, and 1 μg/mL, separately. For colorimetric assay, a mixture of test solution (2 mL), ethanol (20 mL), and ethanolic solution of aluminum chloride (5%, 1 mL) were all mixed, kept for 30 min, and the absorption was measured at 425 nm by a UV–Vis double beam equipment (Analytik Jena, SPECORD 210 PLUS, Jena, Germany). The experiment was performed in triplicate in three consequent days. The output data showed proper linearity (R2 = 0.9976) and proper inter- and intra-day precision and accuracy in the range of 1 μg/mL to 50 μg/mL. FABRICATION OF NANOPARTICLES In this study, nanoparticles were prepared by O/W emulsification and solvent evaporation method (27). Some preliminary studies were performed for optimization of nanoparticles (data not shown). All experiments were done on triplicate.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)