38 JOURNAL OF COSMETIC SCIENCE The optimized nanoparticles were prepared as follows: Firstly, 50 mg of PLGA was dissolved in dichloromethane (1.5 mL). Then 1 mL of the ethanolic extract of propolis (equivalent to 5.5 mg/mL of galangin) was added to the clear solution. The organic phase was heated on a water bath to 50°C and stirred on a stirrer (IKA®-Werke GmbH, Staufen, Germany) at 500 rpm. The aqueous phase was prepared by dissolving polyvinyl alcohol (2%) and was heated on a water bath to reach the equal temperature of the previously prepared organic phase. Then, the organic phase was added dropwise into the aqueous phase under a homogenizer mixer (at 15,000 rpm), and the residual organic solvent was evaporated using a Heidolph® rotary evaporator (Heidolph Instrument GmbH, Schwabach, Germany). For precipitation of nanoparticles, the prepared opalescent colloidal sample was centrifuged at 14,000 rpm for 30 minutes at 4°C. Then, the settled-down nanoparticles were collected for further experiments while the transparent aqueous phase was collected for determination of EE% and loading efficiency percentage (LE%). For better comparison, the plain PLGA nanoparticles, which did not contain PFs, were fabricated and characterized by the same procedures. CHARACTERIZATION OF THE PHYSICOCHEMICAL PROPERTIES OF NANOPARTICLES Determination of size, PdI, and zeta potential. For in vitro characterization, the settled-down nanoparticles were dispersed in deionized water and their zeta potential, particle size, and PdI were measured by dynamic light scattering technique using a nano-sizer (Nano ZS90 Malvern®, Malvern Instruments, Malvern, Worcestershire, United Kingdom) at the wavelength of 415 nm at 25°C with detection angle of 90°. Determination of EE% and LE%. The indirect method was used for determination of EE% and LE% of the nanoparticles. Briefly, the transparent aqueous phase, which was obtained following centrifugation of the freshly prepared colloidal suspension, was analyzed for determination of the amounts of free PFs that were not incorporated in structure of the nanoparticles. The previously mentioned method of aluminum chloride was used for quantification of PFs (based on the amount of galangin) in the aqueous phase. Then the EE% and LE% of the PFs, encapsulated into PLGA nanoparticles, were calculated using following equations [Equation 1, Equation 2]: EE% of initiqal PF Mass of free PF in aqeous solution) = - (Mass ×100 Mass of initial PFs (1) LE% of initial PF Mass of free PF in aqeous solution) = - (Mass ×100 wheight of nanoparticles (2) FREEZE-DRYING OF THE NANOPARTICLES For lyophilization, the nanoparticles were reconstituted using mannitol (1% w/v, 1 mL) as the cryoprotectant and were freeze-dried by an Operon® freeze dryer (Operon, Gyeonggi-do, South Korea) for a period of 48 hours. For determination of the effects of lyophilization process on physicochemical properties of the nanoparticles, the freeze-dried samples were
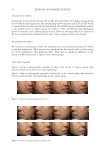
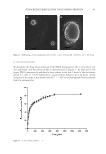
39 SUNSCREEN FORMULATION CONTAINING PROPOLIS then resuspended in double distilled water and their size, PdI, and zeta potential were determined. The data were statistically compared with the corresponding data achieved from nonfreeze-dried samples. The experiments were performed on triplicate. MORPHOLOGICAL STUDIES For investigation of the morphology, the lyophilized samples were examined by scanning electron microscopy (SEM). The nanoparticles were set up by aluminum stubs, coated by a gold layer, and were examined using an SEM (JEOL-JSM-6360JAPAN). The SEM images were acquired in two magnifications of ×4,000 and ×20,000, separately. IN VITRO RELEASE STUDY In this study, diffusion through a dialysis bag was used to investigate the release profile of PFs from the PLGA nanoparticles (20). Briefly, the proper amounts of the lyophilized nanoparticles, containing PFs equivalent to 4.96 mg of galangin, was resuspended in a phosphate buffer (pH = 5.0). Then after, the colloidal mixture was inserted into a dialysis bag (Molecular cut off 12,000 Da). The bag was then immersed in 150 mL of a phosphate buffer (pH = 5.0) mimicking the skin condition. For increasing the solubility of PFs, TWEEN 80® (Croda Americas, Inc., Princeton, NJ, United States 2% w/v) was added to the release medium. The volume of the medium was selected in a manner that establishment of the sink condition was ascertained. The medium was shaken at 150 rpm while the temperature was kept constant at 32 ± 1°C (mimicking the skin temperature) using a Memmert® shaker incubator (Memmert GmbH, Schwabach, Germany). At different time intervals, 1.5 mL of the medium was collected and was immediately replaced by the equal volume of previously heated fresh buffer. The amounts of galangin in the collected samples were determined spectrophotometrically, and finally the cumulative release profile of galangin, as the representative of PFs, was calculated. FORMULATION OF THE SUNSCREEN For evaluation of efficacy of the nanoparticles as a sunscreen agent, the nanoparticles were incorporated into a sunscreen formulation. For this purpose, a pharmaceutical O/W placebo cream base was formulated. Briefly, suitable amounts of cetosrearyl alcohol (6% w/w) were melted in a stirrer, equipped with a hot plate. The molten was mixed with proper amounts of liquid paraffin (5% w/w) and isopropyl myristate (5% w/w). Then, the prepared oil phase was heated to the temperature of 70°C. For preparation of the aqueous phase, proper amounts of Cetomacrogol 1000® (Croda™ International, Snaith, United Kingdom 1.5 w/w) as the emulsifier was dissolved in appropriate volume of deionized water (82.5 w/w) and was heated to the equal temperature of the oil phase. The oil phase was then added gradually to the aqueous phase under stirring and the prepared O/W emulsion was kept nonagitated for a period of 24 hours to form a suitable pharmaceutical cream base. To avoid any bubble formation, the mixing process was performed in vacuum chamber. The prepared cream was evaluated visually for proper physicochemical properties such as viscosity, thickness, homogeneity, and spreadability. Then, suitable amounts of either the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)