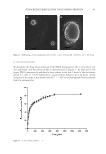
44 JOURNAL OF COSMETIC SCIENCE DETERMINATION OF THE SPF The results for in vitro determination of the SPF of the sunscreen samples are shown in Figure 4. As shown in the figure, sunscreen formulations incorporating propolis-containing PLGA nanoparticles exhibited significantly higher protection compared to other sunscreen formulations (p 0.05). Furthermore, as depicted in the figure, the plain PLGA nanoparticles showed only a slight photoprotection, which was significantly lower than sunscreen formulation containing free propolis. DISCUSSION According to several studies, propolis exhibits various biological properties, and its potential for wide applications in pharmaceutical and food processing industries have attracted much interest (25,28–30). Propolis is a natural substance that contains antioxidant, antimicrobial, and anti-inflammatory compounds, and it can be incorporated in sunscreen formulations as a potential sun-protecting cosmeceutical agent (4). Various studies have identified the structure of different polar and nonpolar compounds of propolis and have characterized their biological properties (24,31,32). The lipophilic nature of the major compounds of propolis causes its low aqueous solubility and therefore the commercial formulations of propolis are mostly based on ethanol, which may be accompanied by local adverse reactions such as neurotoxic effects (33). Nanoparticles are useful drug delivery systems to elaborate the bioavailability of propolis formulations (34). Moreover, nanoparticles could protect propolis from environmental degradation and therefore enhance the stability of the formulations (20,35,36). Encapsulation of the propolis into PLGA nanoparticles has been previously studied and showed enhanced Figure 4. Determination of SPF. ***p 0.001.
45 SUNSCREEN FORMULATION CONTAINING PROPOLIS antimicrobial, antioxidant, and anti-inflammatory properties of the compound (18,20). In this study, the photoprotective properties of PLGA nanoparticles encapsulating PFs were investigated. The nanoparticles were characterized physiochemically and the release profile of PFs from nanoparticles was determined. Finally, the efficacy of the formulations in exerting sun-protecting effects was evaluated. Preliminary studies showed that higher PLGA concentration as well as longer homogenization time could increase the size and PdI of the nanoparticles (35,37). The findings were in accordance with Iadnut et al. (27), which developed propolis-containing PLGA nanoparticles to assess the antifungal activity against Candida albicans. The observed increase in particle size at higher PLGA concentrations may probably be due to increasing viscosity and therefore subsequent poor dispersibility of PLGA into aqueous phase (38). As represented in Figure 3, the in vitro release study revealed a biphasic pattern containing an initial burst release of PFs from the nanoparticles, which was due to release of flavonoids that were accumulated in the surface of particles, followed by a sustained and prolonged release profile in further time intervals, which belonged to the slow diffusion rate of flavonoid molecules that were entrapped into the nanoparticles. Similarly, Derman et al. (39) showed a slow release of caffeic acid phenolic esters, which were encapsulated in the PLGA nanoparticles. In the literature, the sun protection effects of ethanolic propolis extracts have been proven (3,4,17). In this study, determination of SPF using in vitro method revealed higher SPF values of propolis-containing nanoparticles compared to free PFs. It is also suggested that PFs have the potency to ameliorate the UV-induced skin erythema by exerting anti- inflammatory, antioxidant, and ROS scavenging properties (11,12). In agreement with the data, Sahlan et al. (18) showed that encapsulation of propolis in casein micelles could improve the photoprotection activity by increasing the SPF value. Although the sunscreen formulation containing plain nanoparticles exhibited some degree of photoprotection (i.e., the calculated SPF value of 6.4 ± 0.78), but the calculated SPF value was much lower than the appropriate values in free propolis and propolis-containing nanoparticles. The slight photoprotection of the plain nanoparticles can be justified by their opalescent characteristics (40). CONCLUSION In this study, the photoprotection effect of PF-loaded PLGA nanoparticles was investigated, and it was shown that entrapment of PFs into the PLGA nanoparticles can improve their SPF. However, it is interesting to investigate the effects of the prepared nanoparticles on reduction of skin inflammatory biochemical markers such as enzymatic activities of COX-1, COX-2, 5-lipooxygenase, and myeloperoxidase, as well as secretion of PGE2 and TNF-α. Moreover, the effects of the nanoparticles on expression levels of antioxidant enzymes such as glutathione S-transferases A1-2 (GSTA1-2), GSTA3, and GSTA4, and the determination of the ROS activity in skin are yet to be studied. Investigation of the cellular uptake of nanoparticles in skin layer using confocal laser microscopy and determination of the uptake mechanism is also worthy of investigation. Finally, the efficacy of nanoparticles should be determined following clinical studies on human skin.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)