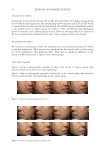
50 JOURNAL OF COSMETIC SCIENCE SKIN AGING There are two types of aging affecting the skin: (1) Intrinsic aging occurs mostly regardless of the levels of exposure of the skin to damaging elements at genetically determined rates and (2) extrinsic aging occurs because of external stressors (e.g., prolonged exposure to sunlight). Table I summarizes the main differences in how extrinsic and intrinsic aging manifest in the skin. BLUE LIGHTS IMPACT ON SKIN IN VITRO STUDIES Cell viability. Opländer studied different wavelengths’ effects on human dermal fibroblasts obtained from mammoplasty or abdominoplasty. Dermal fibroblast cell viability decreased at 410 and 420 nm at 60 and 90 J/cm2 (10,11). At 15 and 30 J/cm2, the reduction in viability was not statistically significant, and at 453 nm and higher, no significant reduction in viability was shown. Cells treated with hydrogen peroxide and blue light reduced cell numbers more than hydrogen peroxide alone, showing that blue light further reduces antioxidant activity of fibroblasts, since the reduction of cell numbers was increased when exposed to blue light. Wavelengths of 453 and 480 nm showed no change. Blue light at wavelengths 412–426 nm was toxic to endothelial cells and keratinocytes in a dose-dependent manner (as noted by Liebmann). Cells were irradiated at 0, 33, 66, and 100 J/cm2 every 24 h for 3 d, then 24 h after the last radiation, and the number of viable cells was measured using staining methods. The number of viable keratinocytes and endothelial cells varied inversely with the level of fluence delivered. Cytotoxicity was shown in both types of cells, although keratinocytes were more resistant. At 453 nm, there is no toxicity present at fluences up to 500 J/cm2, much higher than levels delivered by the sun (12). Melanogenesis. Regazzetti also studied blue light impact melanogenesis. Human foreskin obtained from children with skin types III and IV was used to culture melanocytes. Cells were irradiated with 415 nm blue light at 50 J/cm2. Using Western blot to test for proteins, there has been an increased calcium flux with blue light irradiated NHMs, and upregulation of pCAMKII. CAMKII activates CREB, then ERK, and p38 (13). This then phosphorylates melanogenesis-associated transcription factor (MITF), which then Table I A Summary of Aging Events for Extrinsic Versus Intrinsic Aging Types Skin layer Extrinsic aging Intrinsic aging Dermis Flattening of the papillary dermis Abnormal, amorphous, thickened elastic structures Does not show thinning Flattening of papillary dermis Decrease of elastin in papillary dermis Increase of irregular and fragmented elastin in reticular dermis Decreased collagen Thinning Epidermis Thickening (5,6), increases in melanocytes (7) Disorderly maturation of keratinocytes (6) No thickening (6), decrease in melanocytes (7) Reduction in proliferating keratinocytes (7) Stratum corneum (SC) Thickening of the SC (5) Thickening of the SC (5) Skin mechanics Reduced ability to resist and recover from applied stress (8) Reduced ability to resist and recover from applied stress (8) Appearance Deep wrinkles (9,6), age spots (8,9) Fine wrinkles (9,6), age spots (8,9)
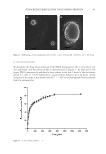
51 POTENTIAL BLUE LIGHT EFFECTS ON AGING activates melanogenesis enzymes, tyrosinase, and dopachrome tautomerase (DCT) (13). Levels of MITF, actin (which aids in melanin transport), DCT, and tyrosinase were tested in irradiated cells against controls immediately as well as 3 d following irradiation, which indicate melanogenesis activity. Actin and MITF were shown immediately after irradiation tyrosinase, DCT, actin, and MITF were present 3 days after irradiation. Reactive oxygen species. NRF2 is a transcription factor that responds to cellular stress such as reactive oxygen species (ROS) and UV light. Its accumulation indicates stressful conditions. It can signal antioxidant response systems to neutralize ROS and other harmful elements. It signals the upregulation of phase 2 antioxidant responders (e.g., glutathione, heme oxygenase, and peroxiredoxins) and UVA light is known to activate NRF2. In a study using human epidermal carcinoma cells injected into mouse skin irradiated with blue light from 400–500 nm, NRF2 was shown to be activated post- irradiation with blue light. Mitochondrial activity decreased beginning at a fluence of 15 J/cm2 and had an 80% reduction at 45 J/cm2 after 72 h of exposure (14). This indicates that there is an increase in ROS activity, and therefore a response in antioxidant systems in the skin. Oxidation events in the skin can contribute to damage in lipid and protein structure, leading to decreased tissue integrity. Inflammatory cytokines. Liebel studied normal human neonatal keratinocytes to analyze free radical production induced by visible light 400–700 nm at 65–180 J/cm2. Liebel exposed these keratinocytes to TNF a , UV (290–400 nm), and either 65, 130, or 180 J/cm2 of visible light, and tested the cells for markers of MAPK/ERK using Western blot. Increased phosphorylation of the epidermal growth factor, detected as phosphor-tyrosine, and the downstream marker of proliferation p42/44 MAPK was detected in the cells treated with TNF a , UV light, and visible light. This suggests activation of the MAPK/ERK pathway (2). Pretreatment of the cells with Tyrphostin, an EGFR inhibitor, blocked the downstream effects on ERK. This suggests that MAPK/ERK resulted from the activation of EGFR. The author concludes that visible light can induce MAPK/ERK in keratinocytes similarly to UV light. There was an increase in ROS formation, IL-1a, MMP1, and MMP9 levels, and in the presence of TNFa, an increase of markers of MAPK/ERK pathway activation. However, these levels only reached the levels of UVA/UVB when exposed directly to levels that exceed those delivered by normal sunlight (2). Liebel’s study on free radical production induced by visible light 400–700 nm at 65–180 J/cm2 was continued using human epidermal equivalents. Cytokine levels were calculated from testing the maintenance medium of the human epidermal equivalents 24 h post-irradiation by immunoassay multiplex kits. The level of MMPs and ROS became significantly increased beginning at 65 J/cm2, while the level of IL-1a became significantly increased beginning at 130 J/cm2 (2). IN VIVO STUDIES Nitric oxide. NO is a signaling molecule in human skin that can also lead to vasodilation. In addition, NO combines with ROS to form peroxynitrite, which is capable of producing lipid peroxidation and damage (15). This can deplete endogenous antioxidant systems in the skin and lead to damage of moisture-enhancing skin lipids, leading to decreased moisture content. In humans, enzymatic NO is produced by dermal cells. Mitochondrial respiration
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)