52 JOURNAL OF COSMETIC SCIENCE can be downregulated by NO (13). Some of the NO molecules formed remain close to their origin as nitroso compounds (e.g., S-nitrosothiols), mercuric chloride nonsensitive nitroso compounds, or the oxidation products nitrite and nitrate. It is known that these derivatives can begin decomposing when exposed to UVA to form bioactive NO. Several studies looked at whether blue light could affect NO release. Liebmann used keratinocytes and epithelial cell cultures in vivo, and irradiated them at 412, 419, and 426 nm at 66–100 J/cm2, and at 453 nm at 500 J/cm2. (12) As shown in Liebmann’s study, adenosine triphosphate levels also increased in keratinocytes and epithelial cells at 453 nm 500 J/cm2 (12). At longer wavelengths of blue light, such as 470 nm, mitochondrial respiration is reactivated. Liebmann estimates that this increase in adenosine triphosphate level is due to the release of NO from NO-sensitive complexes of the respiratory chain, such as bovine serum albumin, which act as photo acceptors, as a result of longer wavelengths of blue light. Liebmann used neonatal keratinocytes as well as reconstituted human epidermal tissue to study the effects of blue light on ROS and cytokines. These cells were exposed to differing doses of blue light ranging from 412–453 nm, UVA at 30 J/cm2, 412 nm at 33 J/cm2, 426 nm at 66 J/cm2, and 453 nm at 100 J/cm2. Blue light was found to have no effect on IL-8 release of human keratinocytes and endothelial cells (12). However, this study also showed that keratinocytes exposed to UVA also did not show an upregulation of IL-8 levels only in endothelial cells did this increase for UVA. IL-8 release can lead to a positive feedback loop and upregulation of elastase, leading to dermal matrix breakdown (15). This study suggests that neither blue light nor UVA light contributes to extrinsic aging from the single cytokine IL-8. For Opländer’s in vivo studies, volunteers were irradiated with a blue light dose of 30 J/ cm2 for 15 min. Opländer also used a closed chamber to collect NO gas from the forearm of human volunteers. An irradiation of 453 nm produced a small increase in gaseous NO emanating from the skin. Wavelengths in the green and red region did not produce a difference in NO levels. 420 nm, 52 J/cm2 produced a slight increase in S-nitroso-protein in homogenates of human skin. Cutaneous blood flow was analyzed with a micro-lightguide spectrophotometer. This resulted in greater NO formation than in vitro studies of human keratinocytes and resulted in NO-induced changes such as dilation of arteries and decrease in blood flow (10,11). There is a copper-dependence to the NO formation. A solution of nitrite-containing 10 mM NaNO 2 , as well as bivalent copper ions 2 mM CuCl 2 , were irradiated with blue light emitting LEDs (420 nm, 453 nm, 50 mW/cm2). Only the copper solution produced NO and did not produce NO with addition of a copper chelator. NO production was linear as a function of irradiance at 453 nm, and NO production peaked at 420 nm (10,11). Inflammatory cytokines. Kleinpenning studied the buttocks skin of eight females of Fitzpatrick skin types I–III with an average age of 20.9, via biopsies following exposure to 20 J/cm2 blue light at 420 nm. The skin was tested for p53, deformed elastin, MMP1, hyperpigmentation, inflammatory cells, keratinocytes, and sunburn cells. MMP1 was found to be present in five volunteer slides but was not significantly different than nonirradiated slides. The level of inflammatory cytokines also did not show a significant increase in irradiated slides from the controls. The author specified that, although blue light does not appear to contribute to extrinsic aging from acute exposure, no conclusions could be drawn from this study about the long-term effects of blue light exposure by the results of this study (19).
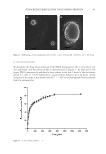
53 POTENTIAL BLUE LIGHT EFFECTS ON AGING Liebel’s study on the effect of visible light on ROS continued with human subjects. The foreheads of volunteers were exposed to visible light 400–700 nm at 50 J/cm2, and ROS was measured by a hydrogen peroxide-sensitive fluorescent probe. Free radical production was shown to increase over the baseline by 85.5% (2). Falcone performed a study to measure effects of 453 nm blue light on healthy skin perturbed using tape stripping. Included were 22 healthy volunteers with a mean age of 21.9 and Fitzpatrick skin types I, II, or III. No toiletries were used 24 h prior, and no sunbathing was permitted 2 weeks prior to experiment. Histamine iontophoresis and tape stripping was performed on two separate spots 4 cm apart on the volar forearm. Irradiance was delivered in pulsed (PW) or continuous (CW) mode. 15 cm of perturbed area of volar forearm was then irradiated with blue light at 453 nm with a fluence of 18 J/cm2 at 10 mW/ cm2 for 30 min. The pulsed mode delivered the same fluence but included a peak irradiance of 200 mW/cm2. Skin surface temperature was also taken from thermocouples of either side of the irradiance area. Six tests were performed over the course of 2 weeks (16). Skin reaction measured by transepidermal water loss (TEWL), skin surface biomarkers interleukin (IL)-1α, IL-1RA, human beta-defensin (hBD)-1, and hBD-2 were measured by means of transdermal analyses patch, and reflectance confocal microscopy was used to measure SC and epidermal thickness. All biomarkers IL-1α IL-1RA, ratio IL-1RA/IL-1α, hBD-1, and hBD-2 increased 24 h after tape stripping during control week. All biomarkers, except IL-1a, increased during irradiation week in the group that was perturbed and irradiated compared to nonperturbed and nonirradiated group. The authors conclude that blue light 453 nm light at 18 J/cm2 results in an inhibitory effect on IL-1a, which will lead to a reduction in release of lamellar bodies, as IL-1a promotes lipid and lamellar body synthesis. This difference was not statistically significant when perturbed and the irradiated group was compared to perturbed nonirradiated group. At 24 h, TEWL was increased, which the authors suggest is due to the reduced IL-1a levels after irradiation and is the first step in the inflammatory cascade, typically increased following barrier disruption (16). At 72 h, no difference in TEWL and epidermal thickness was noted in comparison of irradiated group and control group, suggesting no antiproliferative effect of single treatment. Reactive oxygen species. ROS can cause damage to lipids and proteins and can signal release of inflammatory cytokines. Nakashima performed an in vivo study looking at red, blue, and UV light using reduction-oxidation sensitive green fluorescent protein (roGFP), a fluorescent protein which can be oxidized by glutathione disulfide (GSSG), which is the oxidized form of cellular glutathione, a major redox protection system in human cells (3). If roGFP is present in the cells, it indicates that glutathione is responding to oxidative stress in the cells. Generation of oxidative species results in damage to skin lipids (17) and signal release of inflammatory cytokines by keratinocytes. roGFP was used due to its change in absorption upon oxidation, so quantification was simplified. Two species of mice were used—one expressed the roGFP in mitochondria, and the other expressed the roGFP in the cytosol and nucleus. Human subjects were also used and experiments performed were conducted within the parameters of the Declaration of Helsinki. The hand of participants was irradiated for 10 min at wavelengths of 400–480 nm at fluences of 11 mW/cm2. Blue light caused oxidative response via glutathione cascade in mitochondrial roGFP1 cells, as found by autofluorescence measurements, but not cytosol-based roGFP1 cells as shown in the mouse study.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)