205 Polyphenol-metal Complex With Dopamine
Technology Company Limited, Shanghai, China). The measurements were repeated three
times in different areas of the hair sample. The averages of the measured parameters (L*,
a*, b*) represent the averages of three repeated measurements in different areas. Color
difference (ΔE) is calculated by the formula: ΔE =[(100– L*)2 +(a*)2 +(b*)2]1/2.
COLOR DURABILITY TEST
The dyed hair samples were completely immersed in 50 mL of shampoo solution, soaked,
and rubbed for 1 minute, then dried with a hair dryer for one wash. The washing procedure
was repeated to determine the color parameters for 1, 10, 20, and 40 washes, allowing the
determination of the color durability of the dyed hair samples.
HAIR DYE UV-VIS, FT-IR ANALYSIS, AND SEM CHARACTERIZATION OF HAIR SAMPLES
The UV-visible absorption spectra of GA, DA, GA-Fe(II)+DA were determined using a
UV-visible spectrophotometer (Shanghai Metash Instruments Co., Ltd., UV-5500PC). The
infrared spectra of dopamine hydrochloride, GA, ferrous chloride tetrahydrate, GA-Fe(II)
solution, and GA-Fe(II)+DA solution were determined by FT-IR infrared spectrophotometer
(PerkinElmer, Spectrum Two). The hair morphology was observed using a field emission
SEM Zeiss, Sigma 500).
HAIR STRETCHING TEST
Hair’s tensile strength was measured using an electronic universal material testing
machine (Shimadzu, AGS-X50N AGS-10KN) with a 200 mm scale and a stretching speed
of 20 mm/min.
RESULTS AND DISCUSSION
POLYPHENOL STAINING EFFECT AND CHELATION RATE
Phenolic compounds contain many dihydroxyphenyl or trihydroxyphenyl groups with
high π electron content and aromatics, which provide an effective negative binding site for
cation formation cation-π interactions. These interactions, which dominate non-covalent
cation-π interactions in the presence of other competing motifs, form stable polyphenol-
metal structures. During staining, polyphenol-Fe(II) first comes into contact with the
outer layer of hair scales and then develops color by adsorption, diffusion to the cortex,
and accumulation in its interior to form a polyphenol-Fe(II) network structure. Different
polyphenols react with ferrous ions to form structures that result in different colors and
varying dyeing effects on white hair. We selected eight polyphenol materials, dopamine,
and polydopamine to stain commercial gray hair and measured the color difference value
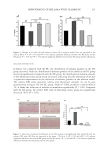
(Figure 2A). In addition, we used ultraviolet spectrophotometry to establish a standard
curve (Figure 2B), measured the absorbance value (A) of each polyphenol material chelated
to Fe(II) at 510 nm, and calculated the chelation rate according to the standard curve and
equation (1)(2)(3)(4)(5) to further screen for the best polyphenol materials.
Technology Company Limited, Shanghai, China). The measurements were repeated three
times in different areas of the hair sample. The averages of the measured parameters (L*,
a*, b*) represent the averages of three repeated measurements in different areas. Color
difference (ΔE) is calculated by the formula: ΔE =[(100– L*)2 +(a*)2 +(b*)2]1/2.
COLOR DURABILITY TEST
The dyed hair samples were completely immersed in 50 mL of shampoo solution, soaked,
and rubbed for 1 minute, then dried with a hair dryer for one wash. The washing procedure
was repeated to determine the color parameters for 1, 10, 20, and 40 washes, allowing the
determination of the color durability of the dyed hair samples.
HAIR DYE UV-VIS, FT-IR ANALYSIS, AND SEM CHARACTERIZATION OF HAIR SAMPLES
The UV-visible absorption spectra of GA, DA, GA-Fe(II)+DA were determined using a
UV-visible spectrophotometer (Shanghai Metash Instruments Co., Ltd., UV-5500PC). The
infrared spectra of dopamine hydrochloride, GA, ferrous chloride tetrahydrate, GA-Fe(II)
solution, and GA-Fe(II)+DA solution were determined by FT-IR infrared spectrophotometer
(PerkinElmer, Spectrum Two). The hair morphology was observed using a field emission
SEM Zeiss, Sigma 500).
HAIR STRETCHING TEST
Hair’s tensile strength was measured using an electronic universal material testing
machine (Shimadzu, AGS-X50N AGS-10KN) with a 200 mm scale and a stretching speed
of 20 mm/min.
RESULTS AND DISCUSSION
POLYPHENOL STAINING EFFECT AND CHELATION RATE
Phenolic compounds contain many dihydroxyphenyl or trihydroxyphenyl groups with
high π electron content and aromatics, which provide an effective negative binding site for
cation formation cation-π interactions. These interactions, which dominate non-covalent
cation-π interactions in the presence of other competing motifs, form stable polyphenol-
metal structures. During staining, polyphenol-Fe(II) first comes into contact with the
outer layer of hair scales and then develops color by adsorption, diffusion to the cortex,
and accumulation in its interior to form a polyphenol-Fe(II) network structure. Different
polyphenols react with ferrous ions to form structures that result in different colors and
varying dyeing effects on white hair. We selected eight polyphenol materials, dopamine,
and polydopamine to stain commercial gray hair and measured the color difference value
(Figure 2A). In addition, we used ultraviolet spectrophotometry to establish a standard
curve (Figure 2B), measured the absorbance value (A) of each polyphenol material chelated
to Fe(II) at 510 nm, and calculated the chelation rate according to the standard curve and
equation (1)(2)(3)(4)(5) to further screen for the best polyphenol materials.